Connecting Science to Practice
Despite the widespread use of gemcitabine and nab-paclitaxel, uncommon
adverse events (AEs) with these drugs, such as myositis, are
often not recognized. In this article, 2 case reports of myositis
linked with nab-paclitaxel treatment in patients with pancreatic
cancer are described. Myositis was identified via a clinical presentation
of focal muscle pain and tenderness, imaging findings, biopsy,
resolution of the myositis after receiving a short course of steroids,
and lack of recurrence after discontinuation of nab-paclitaxel and
the continuation of single-agent gemcitabine. Awareness of myositis
as a potential AE after treatment with nab-paclitaxel is important,
especially in an era of increasing multidrug combination therapies.
The authors’ findings suggest that the discontinuation of nab-paclitaxel
plus the use of steroids can permit the continuation of safer,
more tolerable therapies, shedding light on additional options for
the care of your patients.
Paclitaxel is an antineoplastic agent in the taxane family that exhibits its action by inhibiting the disassembly of microtubules.1 Paclitaxel enhances the action of tubulin dimers to stabilize the existing microtubules and this, in turn, prevents mitosis and leads to cell death.1 Nab-paclitaxel (Abraxane) is a nanoparticle albumin-bound formulation of paclitaxel.2
Nab-paclitaxel plus gemcitabine is a standard-of-care regimen for the treatment of pancreatic adenocarcinoma, as established by a phase 3 trial (N=861) that identified an improvement in median overall survival with the combination of gemcitabine and nab-paclitaxel compared with single-agent gemcitabine (8.5 months vs 6.7 months, respectively; hazard ratio, 0.72; 95% confidence interval, 0.62-0.83; P<.001).3 Long-term (>3 years) survivors were only included in the nab-paclitaxel plus gemcitabine arm (4%).4 The adverse events (AEs) that are frequently related to treatment with nab-paclitaxel include neutropenia, vomiting, peripheral neuropathy, nausea, diarrhea, edema, alopecia, pyrexia, fatigue, dehydration, rash, and decreased appetite.2
The AEs related to paclitaxel and myositis in previous case reports differ from the AEs in this article.5,6 One report described an 80-year-old woman who had pyomyositis and septic shock after receiving biweekly paclitaxel for 3 months and then presenting with shoulder pain, catecholamine-resistant shock, and bradycardia.5 This patient’s clinical course included high fever, erythema, tenderness, edema in the arm, and necrosis of the right shoulder joint muscles.5 She received antibiotics, and the researchers note that chemotherapy was restarted, but there were no further comments on her response or AEs.5
Another case report noted dermatomyositis in a 57-year-old man with nonmetastatic ductal breast carcinoma.6 This patient received 6 cycles of weekly neoadjuvant paclitaxel and had abdominal pain, diarrhea, asthenia, and rash with periocular erythema and interphalangeal joint involvement.6 He started treatment with topical steroids, and a skin biopsy was conducted.6 No further comment was added regarding muscle involvement or restarting treatment with paclitaxel.6
An additional case report outlined the clinical course of a 58-year-old woman with endometrial cancer who received treatment with paclitaxel and carboplatin and subsequently had pyomyositis that was managed with antibiotics and abscess drainage.7 No further elaboration was added with regard to the continuation of therapy.7 In another case report, a 64-year-old woman who had ovarian cancer received paclitaxel and carboplatin every 3 weeks for 6 cycles and had diffuse muscular weakness (among other symptoms) after 6 months of treatment.8 The emphasis of this case report focused on the symptoms of cutaneous sclerosis that were reported.8 Electromyography evaluation was consistent with myositis, but little comment was made regarding muscle weakness.8
Overall, as noted by the case reports that are outlined, the previous reports included concurrent infection and/or skin manifestations or there was no clear relationship between myositis and the administration of paclitaxel.5-8 There are no previous reports of myositis linked with nab-paclitaxel treatment. Herein, we report 2 cases of patients with pancreatic cancer who had focal muscle pain and weakness after receiving infusions of nab-paclitaxel.
Case Reports
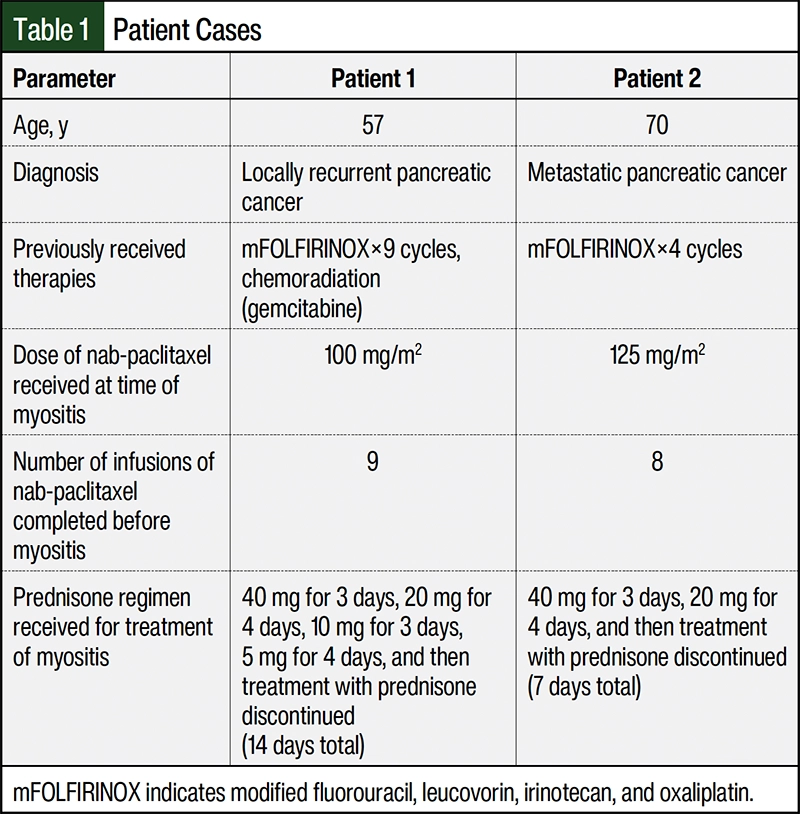
Table 1 includes a summary of the 2 patient cases reported here.
Patient 1
Patient 1 is 57-year-old man with recurrent pancreas cancer. His previous treatment included receiving neoadjuvant modified fluorouracil, leucovorin, irinotecan, and oxaliplatin (mFOLFIRINOX) for 9 cycles, a 5-week course of gemcitabine and radiation followed by distal pancreatectomy and splenectomy for stage ypT3N0Mx invasive ductal adenocarcinoma. Computed tomography surveillance conducted 7 months after surgery identified local tumor recurrence. The patient was initiated on treatment with gemcitabine 1000 mg/m2 and nab-paclitaxel 125 mg/m2 on days 1, 8, and 15 every 28 days.
The day after receiving his ninth infusion of nab-paclitaxel, the patient had a sudden onset of low-grade fever and bilateral anterior thigh pain. The pain was moderate, with palpable tenderness along his anterior thighs. At a follow-up visit 2 weeks later, the symptoms had lessened, although he still had persistent tenderness and pain that was causing mild gait disturbance. A physical examination was notable for palpable tenderness over the bilateral rectus femoris muscles.
The patient’s creatine phosphokinase was 567 IU/L. An ultrasound identified diffuse enlargement of the bilateral rectus femoris muscle bellies, which were heterogeneous in appearance and largely echogenic, with a loss of normal muscle architecture. In addition, there was marked hyperemia within the abnormal muscles on color Doppler interrogation, with abnormal areas that were noncompressible. Subsequent magnetic resonance imaging (MRI) identified long segment diffuse intramuscular edema with associated lacelike postcontrast enhancement involving the bilateral rectus femoris muscles with areas of relative hypoenhancement, which most likely represented myositis. The patient’s medication list was reviewed for agents that could contribute to myositis, with none noted.
Chemotherapy was held, and the patient was prescribed a 14-day course of prednisone (40 mg once daily for 3 days, 20 mg once daily for 4 days, 10 mg daily for 3 days, 5 mg for 4 days, and then discontinued). His symptoms improved and then resolved with this treatment. Treatment with nab-paclitaxel was discontinued, and the patient resumed therapy with gemcitabine and capecitabine with no recurrence of myositis.
Patient 2
Patient 2 was a 70-year-old man with metastatic pancreatic cancer. He previously received 4 cycles of mFOLFIRINOX with documented disease progression. Subsequently, chemotherapy was changed to gemcitabine 1000 mg/m2 and nab-paclitaxel 125 mg/m2 every 14 days. During the fourth month of therapy, swelling, erythema, and discomfort of the left lower extremity and left upper extremity were reported. A deep venous ultrasound scan was negative for deep vein thrombosis (DVT), and the patient received diuretics and oral antibiotics for the treatment of presumed cellulitis without symptom improvement.
Two weeks later, the patient presented to the emergency department with ongoing fatigue, muscle pain, joint pain, weakness, and poor oral intake. He had an occluded biliary stent with gram-negative bacteremia and underwent endoscopic retrograde cholangiopancreatography with stent replacement and intravenous antibiotics. In addition, persistent swelling and pain of the left upper and left lower extremities were investigated. The patient had a creatine phosphokinase test result of 1380 U/L, deep venous ultrasound studies that did not show DVT, and a thigh MRI that showed bilateral interstitial edema of the thigh musculature, with asymmetric involvement of the quadriceps muscle (left<right), which is compatible with extensive myositis with early myonecrosis. An MRI of the left arm revealed nonspecific edema of the left forearm musculature, which was most significant within the flexor compartment. A soft tissue biopsy of the left vastus lateralis showed skeletal muscle with patchy necrosis and focal myositis. In response to this finding, ongoing treatment with fenofibrate, atorvastatin, and amlodipine were discontinued.
After the biopsy, the patient received a prednisone taper (40 mg for 3 days, 20 mg for 4 days, and was then discontinued). Throughout the patient’s hospital admission, his creatine phosphokinase trended down, from 1380 U/L to 474 U/L at 8 days, then to 252 U/L at 2 weeks, and then to 93 U/L at 4 weeks from baseline. Because of the concern that nab-paclitaxel precipitated the patient’s myositis, treatment with nab-paclitaxel was discontinued, and the patient’s cancer therapy was changed to gemcitabine plus capecitabine. At 8 weeks from hospital admission, the patient had muscle pain, his swelling and weakness had resolved, and the myositis did not recur since resuming treatment with gemcitabine.
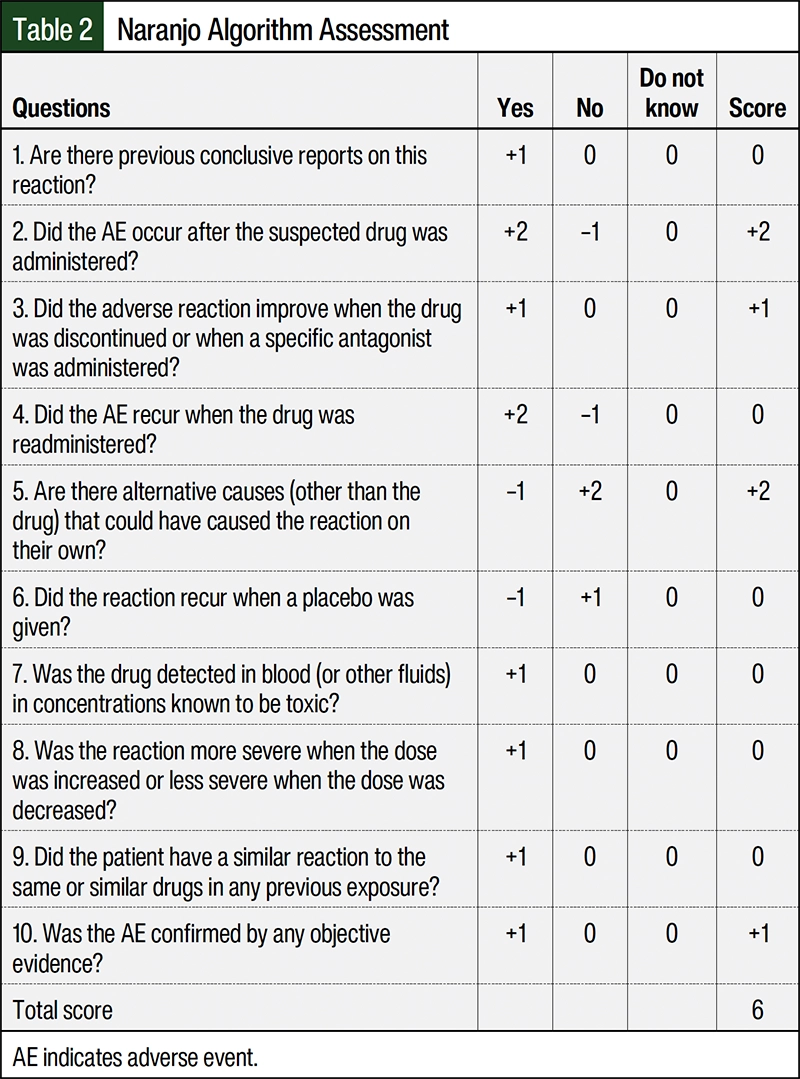
Based on the Naranjo algorithm assessment of our patients’ adverse reactions,9 the final Naranjo score was 6 (Table 2), which suggests that nab-paclitaxel was the probable cause of both of our patients’ symptoms and myositis.
Discussion
Previous case reports of pyomyositis and cutaneous reactions in the setting of treatment with paclitaxel and nab-paclitaxel are different from the myositis that our patients had.5-8 The clinical presentation and course for these 2 patients with pancreatic cancer include focal muscle pain and tenderness, weakness, and imaging compatible with myositis, as well as a biopsy that was confirmed in 1 case, which has not previously been described.
In December 2022, a literature search of the PubMed database using the terms nab-paclitaxel, Abraxane, myositis, and adverse drug reaction revealed no published case reports of nab-paclitaxel–induced myositis in humans. Case reports of paclitaxel-related myositis, septic shock, dermatomyositis, generalized muscle weakness, and pyomyositis were outlined above.5-8 However, none of these reports provided a hypothesized mechanism of these AEs. We believe the rarity of these reports reflects how uncommon myositis is for patients who receive treatment with nab-paclitaxel.
In our 2 case reports, the time frame for the patients’ symptom onset was after 3 to 4 months of nab-paclitaxel therapy, which potentially indicates an idiosyncratic AE as opposed to a sudden, first-exposure hypersensitivity AE. The resolution and recovery from the symptoms of myositis closely followed the discontinuation of treatment with nab-paclitaxel and receiving a short course of prednisone (7-14 days). The half-life of nab-paclitaxel is estimated to be 13 to 27 hours, and, as such, nab-paclitaxel is expected to be clear from the system in approximately 5 days. The use of a steroid taper for longer than this time period helps to reduce the inflammation identified.
Last, when the scoring tool from Naranjo and colleagues9 was applied in our 2 cases, the resulting score was 6. This indicates that a probable drug-related AE resulted from treatment with nab-paclitaxel in our 2 patients. The scoring components were 2 points for the AE after nab-paclitaxel was administered, 1 point for the AE improving with the discontinuation of treatment with nab-paclitaxel, 2 points for the finding of no alternative causes for the AE, and 1 point for the AE being confirmed by objective evidence (by MRI and biopsy in Patient 1 and by ultrasound imaging in Patient 2; Table 2). Neither of the patients were rechallenged with nab-paclitaxel because of concerns of progressive myositis and further complications. Both patients restarted treatment with gemcitabine with no recurrence or worsening of myositis, which suggests that gemcitabine was not the offending agent.
To our knowledge, there are no published case reports of myositis related solely to the administration of nab-paclitaxel. There is no clear etiology for this AE. A limitation of this report is that other possible contributing factors could have led to myositis, including concomitant medications in Patient 2, and a remote effect from previous therapy with mFOLFIRINOX. However, based on the timing and resolution of symptoms with the discontinuation of nab-paclitaxel treatment, the attribution of myositis is most likely to nab-paclitaxel. Myositis is a rare AE of treatment with nab-paclitaxel and should be considered if patients present with sudden or progressive muscle tenderness, pain or weakness, muscle swelling, or gait instability during therapy. The discontinuation of treatment with nab-paclitaxel and a 7-to-14-day course of prednisone is an appropriate management strategy based on the 2 case reports presented here.
Conclusion
Although case reports detailing muscle and infectious complications of paclitaxel have been described, to date there are no reports of myositis with nab-paclitaxel. Focal muscle pain, swelling, and weakness consistent with myositis occurred in 2 patients with pancreatic cancer after treatment with nab-paclitaxel, with the resolution of symptoms after the discontinuation of nab-paclitaxel and a short course of steroids.
Author Disclosure Statement
Dr Laza, Dr McDevitt, and Dr Zalupski have no conflicts of interest to report.
References
- Paclitaxel injection, for intravenous use [prescribing information]. Alembic Pharmaceuticals; October 2022. Accessed December 15, 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=0c12c77c-5fd4-48dc-a318-e96892233992
- Abraxane (paclitaxel) injection, for intravenous use [prescribing information] Abraxis BioScience; October 2022. Accessed December 15, 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=24d10449-2936-4cd3-b7db-a7683db721e4
- Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369:1691-1703.
- Goldstein D, El-Maraghi RH, Hammel P, et al. nab-Paclitaxel plus gemcitabine for metastatic pancreatic cancer: long-term survival from a phase III trial. J Natl Cancer Inst. 2015;107:dju413.
- Paclitaxel. Reactions Weekly. 2017;1646:260.
- Alamón-Reig F, Rizo-Potau D, Laguna JC, Fuertes de Vega I. Paclitaxel-related eruption mimicking dermatomyositis. J Dtsch Dermatol Ges. 2021;19:1074-1075.
- Singh P, Chan W, Blomfield P, McIntosh R. Pyomyositis after chemotherapy for endometrial cancer. Int J Gynecol Cancer. 2010;20:1256-1258.
- Winkelmann RR, Yiannias JA, DiCaudo DJ, et al. Paclitaxel-induced diffuse cutaneous sclerosis: a case with associated esophageal dysmotility, Raynaud’s phenomenon, and myositis. Int J Dermatol. 2016;55:97-100.
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.