Connecting Science to Practice
Vaginal cancer is uncommon, and as a result, treatment
data are limited, with treatment options primarily extrapolated
from cervical cancer. Recently, the National Comprehensive
Cancer Network published guidelines for vaginal
cancer, recommending pembrolizumab for metastatic or
recurrent programmed death-ligand 1–positive tumors. A
treatment option for second-line or subsequent therapy included
tisotumab vedotin, but data evaluating the combination
of tisotumab vedotin and pembrolizumab in vaginal
cancer are lacking, although there are promising results of
this combination in cervical cancer. This case report assesses
tisotumab vedotin and pembrolizumab in recurrent,
metastatic vaginal cancer. The authors determined that it
may be an effective option with tolerable adverse events,
suggesting more research should be done evaluating this
regimen in this rare disease.
Vaginal cancer is a rare cancer that accounts for 1% to 2% of gynecologic malignancies each year.1 Vaginal cancer is most common in older women, with an approximate age of diagnosis of 69 years.1 The prognosis of vaginal cancer is poor, with an estimated overall 5-year relative survival rate of 51%.2 The National Comprehensive Cancer Network (NCCN) guidelines for vaginal cancer recommend chemoradiation for early-stage disease and systemic therapy for recurrent or metastatic disease.3 The preferred first-line therapy for recurrent or metastatic disease depends on programmed death-ligand 1 (PD-L1) expression, and regimens for PD-L1–positive tumors include pembrolizumab in addition to doublet chemotherapy with or without bevacizumab. Subsequent therapy for recurrent or metastatic vaginal cancer includes pembrolizumab for PD-L1–positive tumors, single-agent chemotherapy, or tisotumab vedotin. Tisotumab vedotin is an antibody–drug conjugate that binds to tissue factor–expressing cells and is then internalized and cleaved to exhibit its antimicrotubule effect via monomethyl auristatin E, leading to cell cycle arrest and apoptosis.4 Tisotumab vedotin is approved for the treatment of recurrent or metastatic cervical cancer.4
The innovaTV 204 trial was a multicenter, open-label, single-arm, phase 2 study that enrolled 102 women with recurrent or metastatic cervical cancer who progressed while or after receiving doublet chemotherapy with bevacizumab.5 In this trial, 60% of patients had recurrent disease and received no more than 2 lines of therapy for recurrent or metastatic disease. The patients received 2 mg/kg of tisotumab vedotin (up to 200 mg) intravenously (IV) every 3 weeks until disease progression or an unacceptable adverse event (AE). The objective response rate (ORR) was 24%. Of the 102 patients, 7 (7%) had a complete response and 17 (17%) had a partial response. The median duration of response was 8.3 months, and 62% of the patients had an ongoing response for at least 6 months. At 12 months, the overall survival (OS) rate was 51%. The most common (>20%) AEs during treatment included alopecia (38%), epistaxis (30%), nausea (27%), conjunctivitis (26%), fatigue (24%), and dry eye (23%).5
The innovaTV 301 trial enrolled women with cervical cancer who progressed while or after receiving doublet chemotherapy with bevacizumab and an anti–programmed cell death protein 1 (PD-1) or anti–PD-L1 agent if patients were eligible to receive them.6 The patients received tisotumab vedotin 2 mg/kg IV every 3 weeks or the investigator’s choice of chemotherapy IV until disease progression or an unacceptable AE. The median OS in the tisotumab vedotin group was 11.5 months compared with 9.5 months in the chemotherapy group (P=.004). The ORR was 17.8% versus 5.2% (P<.001), respectively, and the duration of response was 5.3 months versus 5.7 months, respectively. The most common (>30% in either group) AEs in the tisotumab vedotin versus in the chemotherapy group included nausea (33.2% vs 40.2%, respectively), conjunctivitis (31.2% vs 0.4%, respectively), and anemia (23.2% vs 52.3%, respectively).6
Currently, tisotumab vedotin is being studied in combination with pembrolizumab for the treatment of relapsed or metastatic cervical cancer in patients who had not received systemic therapy, excluding chemoradiation.7 Tisotumab vedotin was administered at a dose of 2 mg/kg IV, and pembrolizumab was dosed at 200 mg IV every 3 weeks. A total of 32 patients were included in the interim analysis, and the ORR was 41%. In all, 3 (9%) patients had a complete response and 10 (31%) had a partial response. The median progression-free survival was 5.3 months, and the median OS was not reached. The most common AEs during treatment included alopecia (61%), diarrhea (55%), epistaxis (49%), conjunctivitis (46%), and nausea (46%). Based on these interim results, the use of tisotumab vedotin with pembrolizumab may offer antitumor activity with an expected AE profile.7
This case report presents a patient with metastatic vaginal cancer who received tisotumab vedotin and pembrolizumab. To our knowledge, there is no literature evaluating the use of this regimen for the treatment of vaginal cancer.
Case Report
A 62-year-old woman presented to the emergency department at West Penn Hospital in Pittsburgh, PA, with vaginal discharge, back pain, and bloating in October 2020. Her significant medical history included a hysterectomy as a result of postpartum hemorrhage in 1991. One week after the emergency department visit, the patient was diagnosed with human papillomavirus (HPV)-positive, stage IIB, poorly differentiated squamous cell carcinoma of the vagina. Imaging showed a 1.4×3.2-cm right lateral vaginal cuff mass extending to the middle third of the vagina with possible extension past the vaginal wall. She was initiated on concurrent chemoradiation with cisplatin IV weekly. The patient had tinnitus after 3 treatment cycles with cisplatin and was switched to treatment with carboplatin plus radiation for the remaining 2 cycles, followed by vaginal brachytherapy. In January 2021, imaging of the pelvis showed a decrease in the size of the mass, as well as the right aspect of the vagina with questionable minimal extension into the paravaginal tissues at the 9 o’clock position.
The patient’s vaginal cancer recurred in April 2021 and metastasized to the lungs. She underwent computed tomography (CT)-guided biopsy, which confirmed that she had metastatic squamous cell carcinoma that was consistent with her squamous cell vaginal cancer primary tumor. She then completed 4 cycles of bevacizumab, carboplatin, and paclitaxel, at which point her local disease progressed further. Next-generation sequencing was obtained and showed a tumor mutational burden of 47 mutations per megabase, a tumor proportion score of 20%, and microsatellite stability. No other actionable mutations were identified. The patient’s treatment was changed to pembrolizumab IV every 3 weeks, which she continued to receive for 10 cycles.
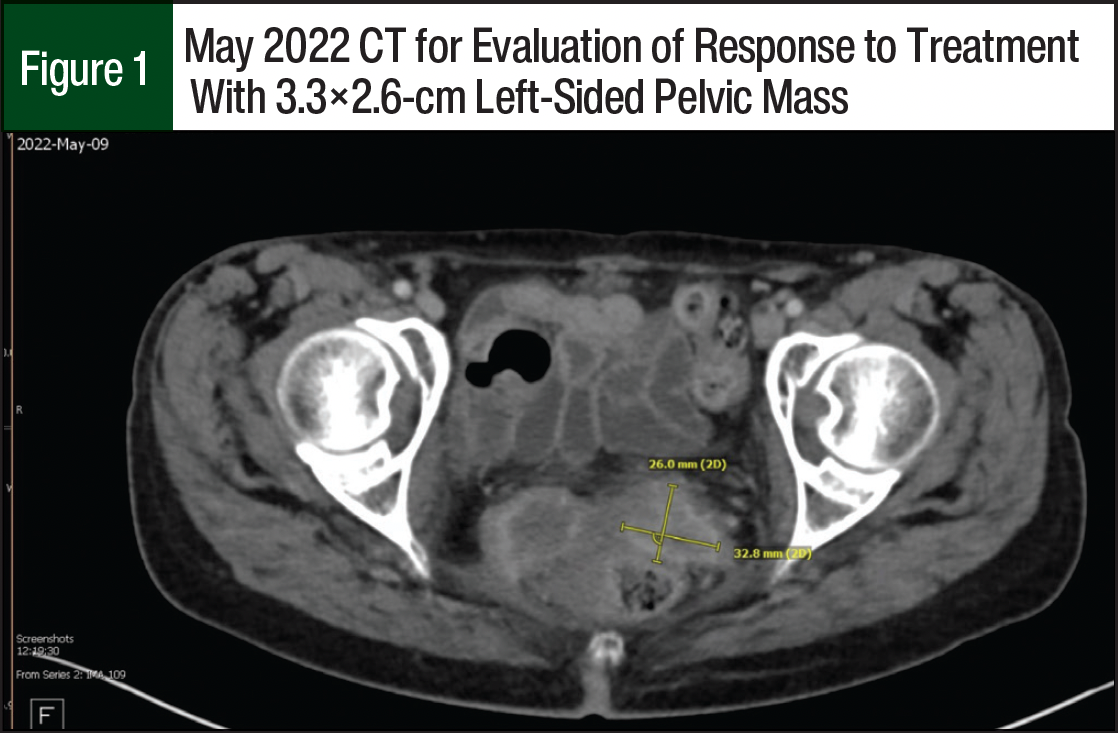
A restaging CT in May 2022 showed small progression of disease with a left-sided pelvic mass measuring 3.3×2.6 cm (Figure 1) and a right-sided pelvic mass measuring 3.7×3.5 cm. The team considered the patient to have stable disease, and the patient received an additional 6 cycles of pembrolizumab. In August 2022, a restaging positron emission testing/CT showed a continued increase in the size of the right-sided pelvic mass now measuring 6.9×4.4 cm; and the pulmonary metastases were not metabolically active. Because of this pelvic disease progression, tisotumab vedotin was added to the current therapy.
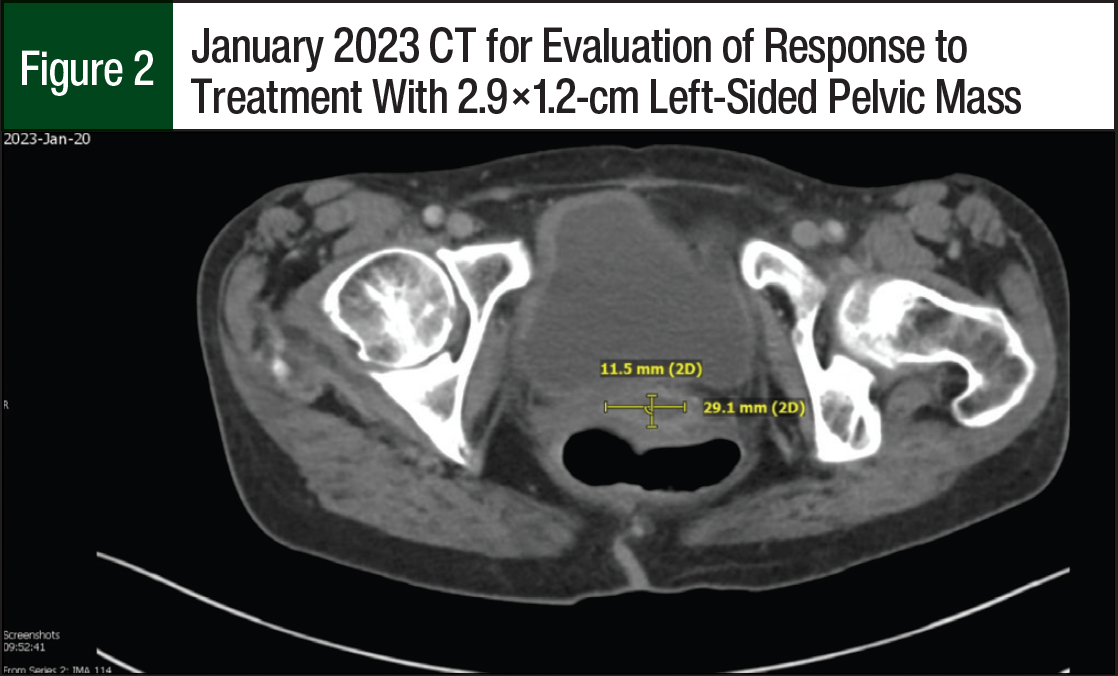
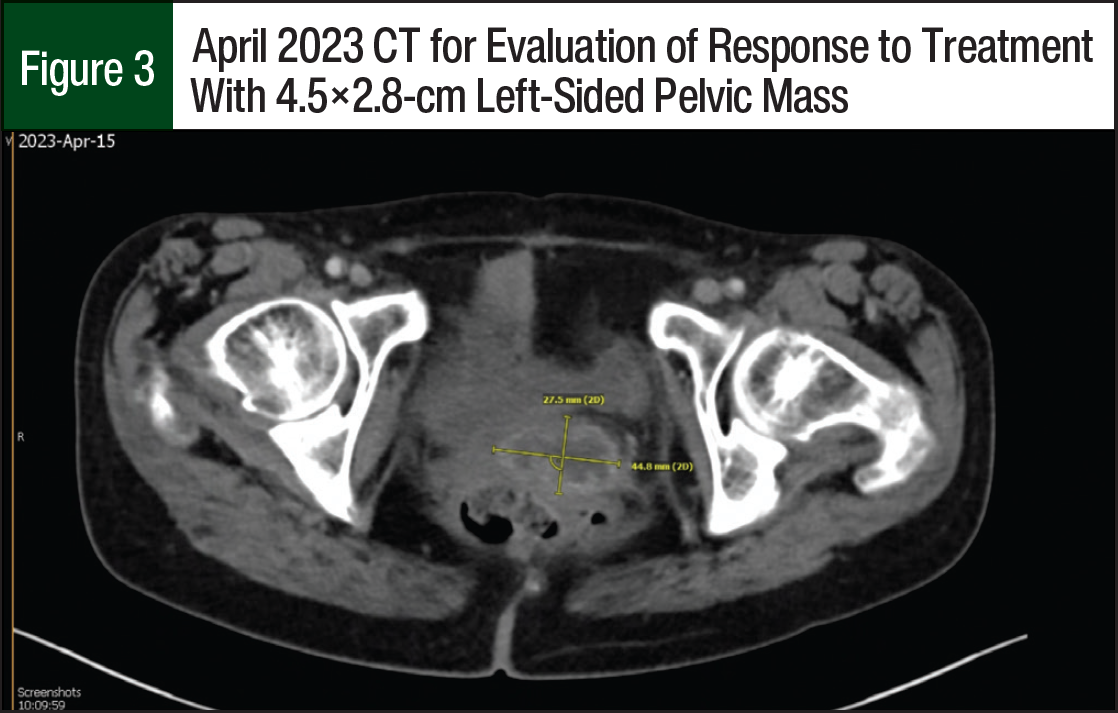
The patient started receiving treatment with tisotumab vedotin 2 mg/kg and pembrolizumab 200 mg IV every 3 weeks. After 6 cycles of the new regimen, a CT scan showed her left-sided pelvic mass decreased in size and now measured 2.9×1.2 cm (Figure 2) and that there was no new metastatic disease. Another CT scan was obtained after an additional 4 treatment cycles, which showed an interval increase in the left-sided pelvic mass that now measured 4.5×2.8 cm (Figure 3), and an unchanged pulmonary nodule signifying that her distant disease appeared to be well controlled. The patient continued systemic treatment and was referred to interventional radiology for pelvic embolization. To date, the patient has received 12 treatment cycles of tisotumab vedotin and pembrolizumab. She has tolerated the regimen well and only had mild dry eye; laboratory testing showed that she also had hypomagnesemia and hypokalemia.
Discussion
Because vaginal cancer is so rare, the treatment options for vaginal cancer have been primarily extrapolated from therapies for cervical cancer. However, the NCCN added a guideline for vaginal cancer in 2024 that provides clearer guidance on recommendations for the treatment of vaginal cancer.3 The NCCN recommends radiation therapy or surgery for stage I disease and chemoradiation with brachytherapy or external beam radiation therapy with brachytherapy for stage II to stage IVA disease. For recurrent or metastatic disease, the preferred first-line therapy includes doublet chemotherapy with or without bevacizumab and the addition of pembrolizumab, a PD-1 inhibitor, to these regimens for PD-L1–positive tumors. For subsequent therapy, the NCCN recommends pembrolizumab as the preferred treatment in patients with PD-L1–positive tumors.3 Other recommended regimens include single-agent chemotherapy, cemiplimab, bevacizumab, or tisotumab vedotin.3,6-9
Data for the treatment of current or metastatic vaginal cancer remain sparse. Platinum-based regimens for vaginal cancer that are extrapolated from treatments for cervical cancer have been used in clinical practice. However, there is growing interest in immunotherapy for patients with relapsed vaginal cancer. It is hypothesized that HPV, which is a risk factor for vaginal cancer, can lead to carcinogenesis because of the body’s inability to clear the virus.8 HPV-driven cancer has a unique microenvironment that helps it evade the body’s immune system. Hypotheses on this mechanism include increased expression of PD-L1, interference with antigen presentation, changes in immune checkpoint receptor interaction, upregulation of cytokines, and alterations in gene transcription.8 Therefore, immunotherapy may be a treatment option. Treatment with pembrolizumab or nivolumab has been studied in patients with recurrent or metastatic vaginal cancer.9,10
How and colleagues presented 2 cases of pembrolizumab monotherapy for vaginal cancer as a case series with data from a phase 2 basket trial.9,11 This trial evaluated treatment with pembrolizumab in patients with rare tumors that are metastatic or not amenable to surgery. In this trial, there were 10 cohorts of advanced, rare tumors, and the 2 cases of vaginal cancer were enrolled in cohort 10 (ie, other rare tumor histologies). Both patients in this case series had recurrent, stage IVB vaginal cancer; were HPV-positive; and had PD-L1–positive expression.9,11
Patient 1 initially had a response to therapy. After 3 cycles of therapy, the target lesion had decreased in size by 68% and continued to decrease with additional treatment cycles.9,11 However, new nodal lesions and progression of nonmeasurable lesions were discovered on imaging after 6 cycles. Ultimately, the patient was administered 10 cycles of single-agent pembrolizumab before treatment was discontinued as a result of disease progression. Patient 2 had no objective response, and the target lesion grew by 71% after 3 treatment cycles. At this time, treatment with pembrolizumab was discontinued. Given the small number of patients with vaginal cancer in this trial, interpretations of these results should be made with caution. Single-agent pembrolizumab was well-tolerated and may have some clinical benefit in patients with recurrent vaginal cancer.9,11
Nivolumab is another PD-1 inhibitor that has been studied in patients with recurrent or metastatic vaginal cancer.10 This phase 1/2 trial enrolled patients with cervical, vaginal, and vulvar carcinoma to receive single-agent nivolumab 240 mg IV every 2 weeks. Of the 24 patients enrolled in the trial, 2 patients had vaginal cancer. The results for patients with vaginal cancer were reported as a composite with patients who had vulvar cancer (N=5). In the vaginal and vulvar cancers arm, the ORR was 20%. None of the patients had a complete response, and 1 patient had a partial response. The 1 patient who responded to treatment had HPV-negative vulvar cancer and had a response to treatment for 5 months. In addition, in the vaginal and vulvar cancers arm, 60% of the patients had stable disease and 20% had progressive disease. None of the median survival data were reported for the vaginal and vulvar cancers arm.10
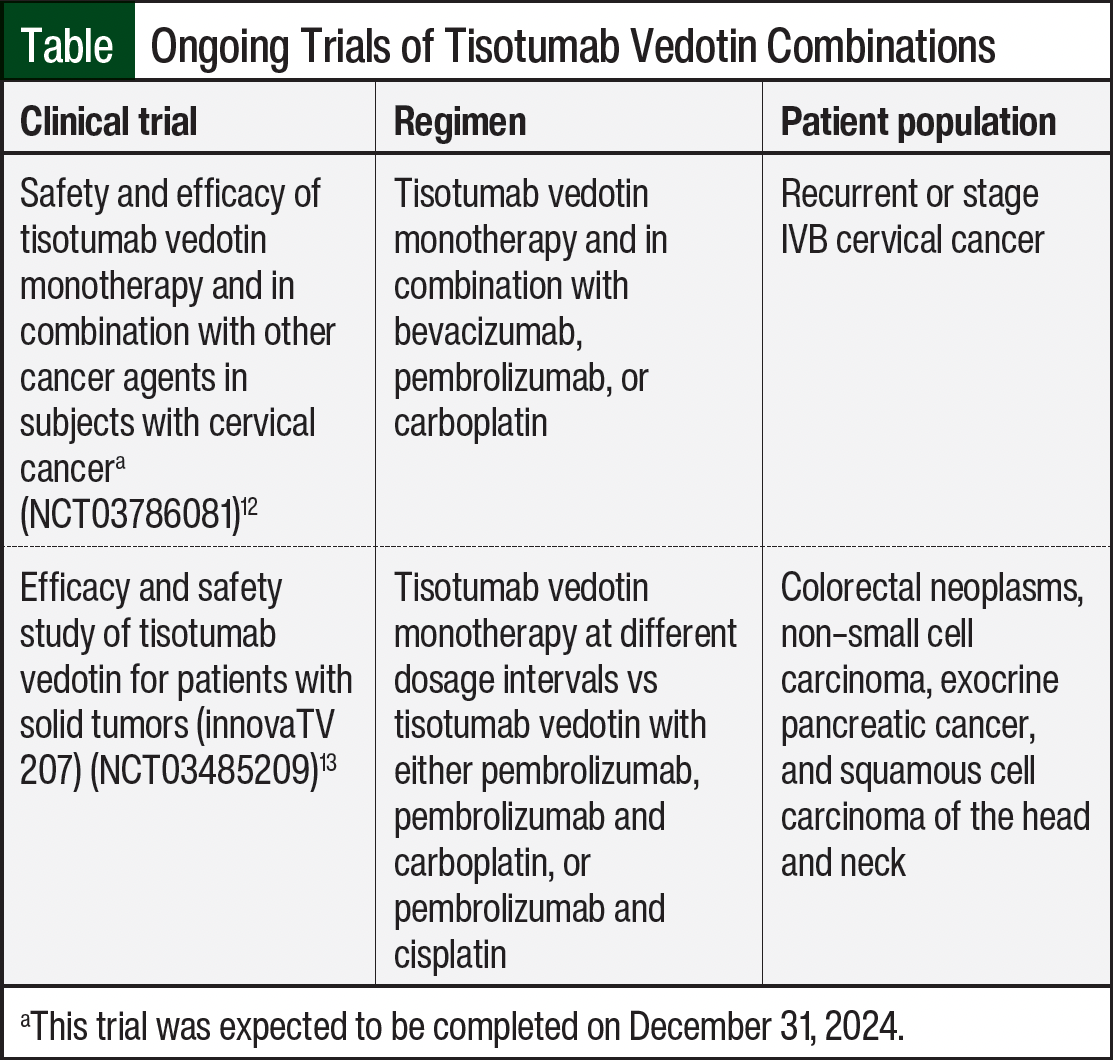
The results from immunotherapy trials are not remarkable; however, there are few reports of the use of immunotherapy given the rarity of this disease. Treatment with tisotumab vedotin plus pembrolizumab has shown promising results in patients with recurrent and metastatic cervical cancer.7 To date, this is the first case report of this regimen being used for the treatment of advanced, metastatic vaginal cancer. In addition, at the time this article was written, there were 2 ongoing trials evaluating treatment with tisotumab vedotin in combination with other agents, one of which was scheduled to be completed on December 31, 2024 (ie, NCT03786081; Table).12,13 The combination of tisotumab vedotin and pembrolizumab has shown promising interim results in the innovaTV 205 trial, and it has been proposed that antibody–drug conjugates work synergistically with immune checkpoint inhibitors, such as PD-1 inhibitors.12,14 One proposed mechanism is a boost in maturation of dendritic cells by the cytotoxic payload of antibody–drug conjugates leading to the recruitment of T cells to the tumor site, potentially enhancing the response to immune checkpoint inhibitors.14
The patient in our case report had an initial response to treatment demonstrated by the decrease in size of the local lesion after 6 cycles. However, after 6 months of treatment (10 cycles total), the patient did have local progression while the distant disease remained controlled. Overall, the regimen has been well-tolerated, with the only AE being dry eye, despite following the eye care recommendations in tisotumab vedotin’s prescribing information.4 Based on the patient’s response, she continued receiving the current regimen with local embolization to the enhancing pelvic lesion.
Conclusion
This case report demonstrates that pembrolizumab and tisotumab vedotin may have activity in the treatment of recurrent and metastatic vaginal cancer. The regimen received by our patient was well-tolerated with no new safety concerns. There is limited guidance for the treatment of advanced and recurrent vaginal cancer.
To date, this is the first report of a patient receiving tisotumab vedotin plus pembrolizumab for the treatment of recurrent vaginal cancer. Future studies evaluating gynecologic cancers, such as cervical or vulvar cancer, should include a cohort for patients with vaginal cancer to better guide healthcare providers in the treatment of this rare disease.
Author Disclosure Statement
Dr Krivak is a consultant, is a researcher for, and is on the Speaker’s Bureau for AstraZeneca, GlaxoSmithKline, Merck, and Myriad, and is a consultant and has been a researcher for Seagen. Dr Hill, Dr Nice, and Dr Moffa have no conflicts of interest to report.
References
- American Cancer Society. Key statistics for vaginal cancer. Updated September 23, 2024. Accessed January 17, 2025. www.cancer.org/cancer/types/vaginal-cancer/about/key-statistics.html
- American Cancer Society. Survival rates for vaginal cancer. Updated September 23, 2024. Accessed January 17, 2025. www.cancer.org/cancer/types/vaginal-cancer/detection-diagnosis-staging/survival-rates.html
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): vaginal cancer. Version 3.2025. December 16, 2024. Accessed January 17, 2025. www.nccn.org/professionals/physician_gls/pdf/vaginal.pdf
- Tivdak (tisotumab vedotin) injection, powder, for solution [prescribing information]. Seagen; April 2024. Accessed January 17, 2025.
- Coleman RL, Lorusso D, Gennigens C, et al. Efficacy and safety of tisotumab vedotin in previously treated recurrent or metastatic cervical cancer (innovaTV 204/GOG-3023/ENGOT-cx6): a multicentre, open-label, single arm, phase 2 study. Lancet Oncol. 2021;22:609-619.
- Vergote I, González-Martín A, Fujiwara K, et al. Tisotumab vedotin as second- or third-line therapy for recurrent cervical cancer. N Engl J Med. 2024;391:44-55.
- Lorusso D, Vergote I, O’Cearbhaill RE, et al. Tisotumab vedotin (TV) + pembrolizumab (pembro) in first-line (1L) recurrent or metastatic cervical cancer (r/mCC): interim results of ENGOT Cx8/GOG 3024/innovaTV 205. Presented at: 2022 ASCO Annual Meeting I; June 3-7, 2022. Abstract 5507.
- Julian R, Savani M, Bauman JE. Immunotherapy approaches in HPV-associated head and neck cancer. Cancers (Basel). 2021;13:5889.
- How JA, Jazaeri AA, Soliman PT, et al. Pembrolizumab in vaginal and vulvar squamous cell carcinoma: a case series from a phase II basket trial. Sci Rep. 2021;11:3667.
- Naumann RW, Hollebecque A, Meyer T, et al. Safety and efficacy of nivolumab monotherapy in recurrent or metastatic cervical, vaginal, or vulvar carcinoma: results from the phase I/II CheckMate 358 trial. J Clin Oncol. 2019;37:2825-2834.
- Naing A, Meric-Bernstam F, Stephen B, et al. Phase 2 study of pembrolizumab in patients with advanced rare cancers. J Immunother Cancer. 2020;8:e000347.
- Safety and Efficacy of Tisotumab Vedotin Monotherapy and in Combination With Other Cancer Agents in Subjects with Cervical Cancer. NLM identifier: NCT03786081. Updated February 12, 2024. Accessed January 17, 2025. https://clinicaltrials.gov/study/NCT03786081
- Efficacy and Safety Study of Tisotumab Vedotin for Patients With Solid Tumors (innovaTV 207). NLM identifier: NCT03485209. Updated November 13, 2024. Accessed January 17, 2025. https://clinicaltrials.gov/study/NCT03485209
- Nicolò E, Giugliano F, Ascione L, et al. Combining antibody-drug conjugates with immunotherapy in solid tumors: current landscapes and future perspectives. Cancer Treat Rev. 2022;106:102395.