Breast cancer is one of the most common cancers globally in women, accounting for approximately 12% of all cancer deaths.1 The most common pathologic subtype is hormone receptor–positive, HER2-negative, which accounts for 68% of female breast cancer cases in the United States.2 Cyclin-dependent kinase (CDK) 4/6 inhibitors have been developed to treat hormone receptor–positive, HER2-negative advanced breast cancer.3 These oral agents have been shown to prolong survival end points and transform the management of advanced or metastatic breast cancer. The transformation led to CDK4/6 inhibitors becoming the preferred first-line therapy in combination with endocrine therapy for this patient population.3
The CDK4/6 inhibitors that are currently approved by the FDA include palbociclib, ribociclib, and abemaciclib.4-6 The results of the PALOMA-3 trial showed a median progression-free survival (PFS) of 9.5 months with palbociclib plus fulvestrant versus 4.6 months with placebo plus fulvestrant (hazard ratio [HR], 0.46; 95% confidence interval [CI], 0.36-0.59; P<.0001).7 The results of the MONALEESA-7 trial demonstrated a median PFS of 23.8 months with ribociclib plus tamoxifen or a nonsteroidal aromatase inhibitor and goserelin versus 13 months with placebo plus tamoxifen or a nonsteroidal aromatase inhibitor and goserelin (HR, 0.55; 95% CI, 0.44-0.69; P<.0001).8 The results of the MONARCH-3 trial showed a median PFS of 28.18 months in the group that received abemaciclib plus an aromatase inhibitor versus 14.76 months in the group that received placebo plus an aromatase inhibitor (HR, 0.54; 95% CI, 0.418-0.698; P=.000002).9 Overall, CDK4/6 inhibitors in combination with endocrine therapy represent a novel treatment option for this pathologic subtype. However, the clinical trial results for all 3 agents also demonstrated adverse events that require close monitoring of patients to avoid treatment discontinuation.7-9
One of the major adverse events of CDK4/6 inhibitors is neutropenia.4-6 For example, grade 3 or 4 neutropenia was reported in 65% of the patients who received palbociclib with fulvestrant in the PALOMA-3 trial.7 Grade 3 or 4 neutropenia was also reported in 61% of patients who received ribociclib in the MONALEESA-7 trial and in 21.9% of patients who received abemaciclib in the MONARCH-3 trial.8,9 Unfortunately, this hematologic adverse event resulted in dosage reductions or treatment delays during therapy.10 For this reason, the close monitoring of the absolute neutrophil count (ANC) is important when patients are receiving CDK4/6 inhibitors, especially during the first 2 months of therapy. The prescribing information for these agents recommends the monitoring of complete blood counts (CBCs) at the beginning of each cycle, as well as every 2 weeks for the first 2 months, and as clinically indicated.4-6
Despite clear guidance on laboratory monitoring for hematologic adverse events, scheduling and follow-up in the initial months of therapy with CDK4/6 inhibitors are complicated by challenges with prescription fulfillment of this costly novel therapy. Oncology clinical pharmacists at St Luke’s Cancer Institute working in a medically integrated pharmacy (MIP) identified an opportunity to expand clinical services and improve the monitoring for neutropenia under their collaborative practice agreement (CPA). When patients initiate treatment with an oral oncolytic agent, oncology clinical pharmacists review the prescriptions, evaluate pertinent laboratory values, and counsel patients about potential adverse events. They will contact the patient every 2 weeks for the first month of treatment to discuss adverse events, such as diarrhea, that could impact the patient’s quality of life. Pharmacists may also order laboratory testing under the CPA to assist providers in monitoring adverse events, such as neutropenia and renal or hepatic dysfunctions. Taraba and colleagues developed and implemented a pharmacist-led virtual clinic that demonstrated improved adherence to laboratory testing in patients who are receiving CDK4/6 inhibitors.11 Similarly, we developed an internal educational sheet, Management of Neutropenia with CDK4/6 Inhibitors, to guide oncology clinical pharmacists in the MIP on the manufacturer-recommended monitoring for neutropenia (Appendix I). During our weekly staff meetings in September 2022, we reviewed this educational guide and emphasized the importance of timely CBC orders when managing patients who are receiving CDK4/6 inhibitors.
The purpose of this article is to evaluate the outcomes of a pharmacist-driven approach in monitoring for hematologic adverse events after receiving CDK4/6 inhibitors in women with advanced breast cancer to identify areas for optimization in clinical practice. Oncology clinical pharmacists who are physically integrated within the clinic collaboratively drive care with providers by providing close patient follow-up, managing adverse events, and optimally timing patients’ treatment schedules. In contrast, prescriptions filled by external sources have minimal-to-no pharmacist oversight, regardless of the risk category. The findings will be reported to leadership in support for additional pharmacist resources in the MIP to provide high-quality patient care and expand clinical documentation specific to CDK4/6 inhibitors in the medical records.
The primary objective of this study was to evaluate laboratory monitoring for CDK4/6 inhibitors according to an internal guidance document for the management of neutropenia via a pharmacist-driven approach compared with patients receiving CDK4/6 inhibitors from external fulfillment. The secondary objectives between the 2 cohorts included assessing the incidence of grade ≥3 neutropenia, the management of grade ≥3 neutropenia, and the average number of days for treatment delays.
Methods
This study was a single-center, prospective analysis of women aged ≥18 years with advanced breast cancer who initiated treatment with a CDK4/6 inhibitor between September 26, 2022, and March 26, 2023. The identification data of patients who received a CDK4/6 inhibitor were extracted from the database chart and were reviewed for the following inclusion criteria: adult women, being diagnosed with breast cancer, and initiating a CDK4/6 inhibitor between September 26, 2022, and March 26, 2023. Patients were excluded from the study if they did not meet the above criteria during the study period or if the pharmacists’ documentation was incomplete. Two patient cohorts were identified, which included those who were receiving CDK4/6 inhibitors from the MIP and were managed by oncology clinical pharmacists in a pharmacist-driven approach, and those who received CDK4/6 inhibitors from external fulfillment sources who were managed in a non–pharmacist-driven approach, such as patient-assistance programs or mail-order pharmacies.
The patients who met the inclusion and exclusion criteria were identified using electronic health records. Data were collected from all of the participants, including age, menopause status (pre-/perimenopause or postmenopause), medical record number, type of CDK4/6 inhibitor received, concurrent hormonal therapy, fulfillment location (MIP, mail order, free drug), start date of CDK4/6 inhibitor, laboratory values, grade of neutropenia, number of dose reductions, treatment delays, and adverse events.
Statistical Analysis
The sample size was determined by the inclusion of the time frame and was not designed to target statistical power. Descriptive statistics were used to report continuous variables as the median values and categorical variables as counts and percentages.
Results
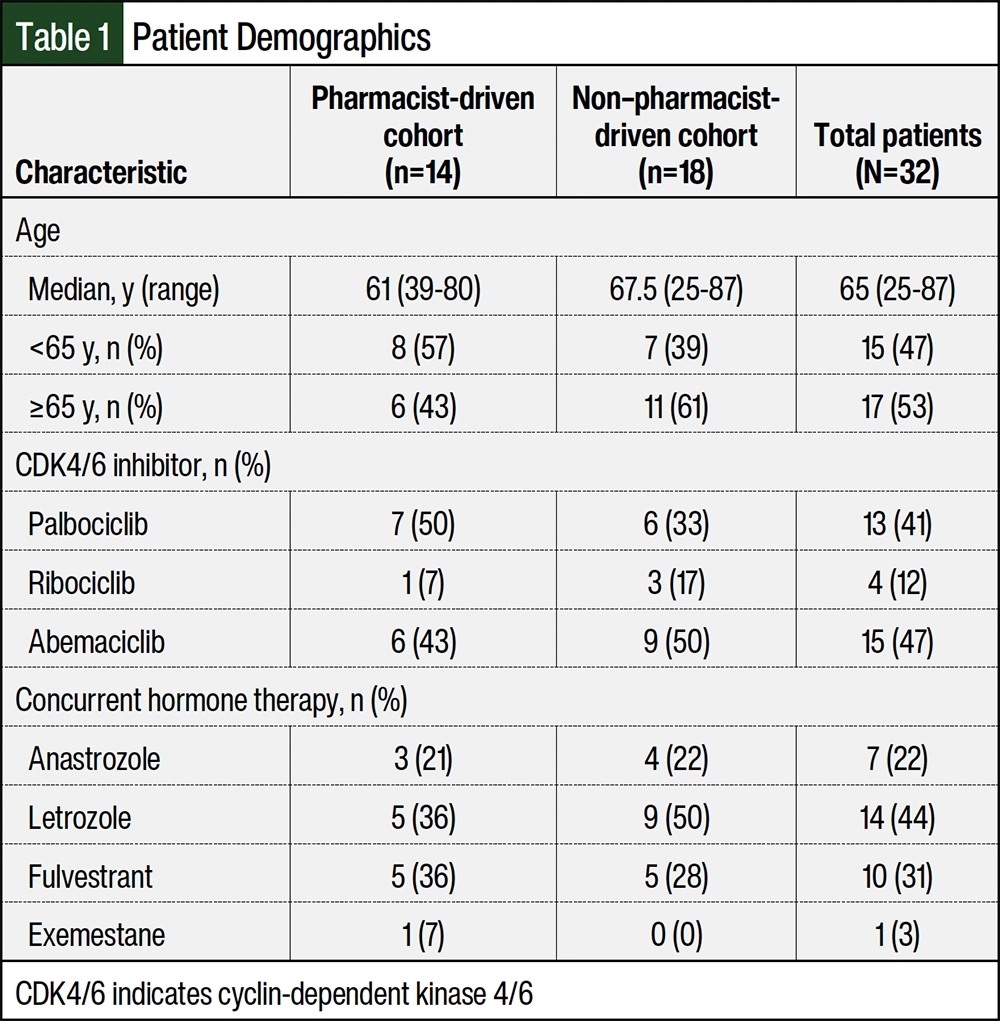
A total of 32 patients met the inclusion criteria during the study’s time frame. Approximately 44% of the patients initiated on a CDK4/6 inhibitor were monitored by oncology clinical pharmacists in the MIP, and approximately 56% of the patients received a CDK4/6 inhibitor through patient-assistance programs or mail-order pharmacies. The baseline characteristics of the representative sample study population (N=32) are shown in Table 1. Most of the new prescriptions were for abemaciclib and palbociclib, which accounted for 47% and 41% of the prescriptions, respectively. The patients’ demographics were similar between the 2 cohorts, except patients in the non–pharmacist-managed group were slightly older than the patients managed by pharmacists. Letrozole was the most frequent concurrent endocrine therapy received with a CDK4/6 inhibitor, accounting for 44% of the patients. Only 1 patient was receiving exemestane as a concurrent hormone therapy (Table 1).
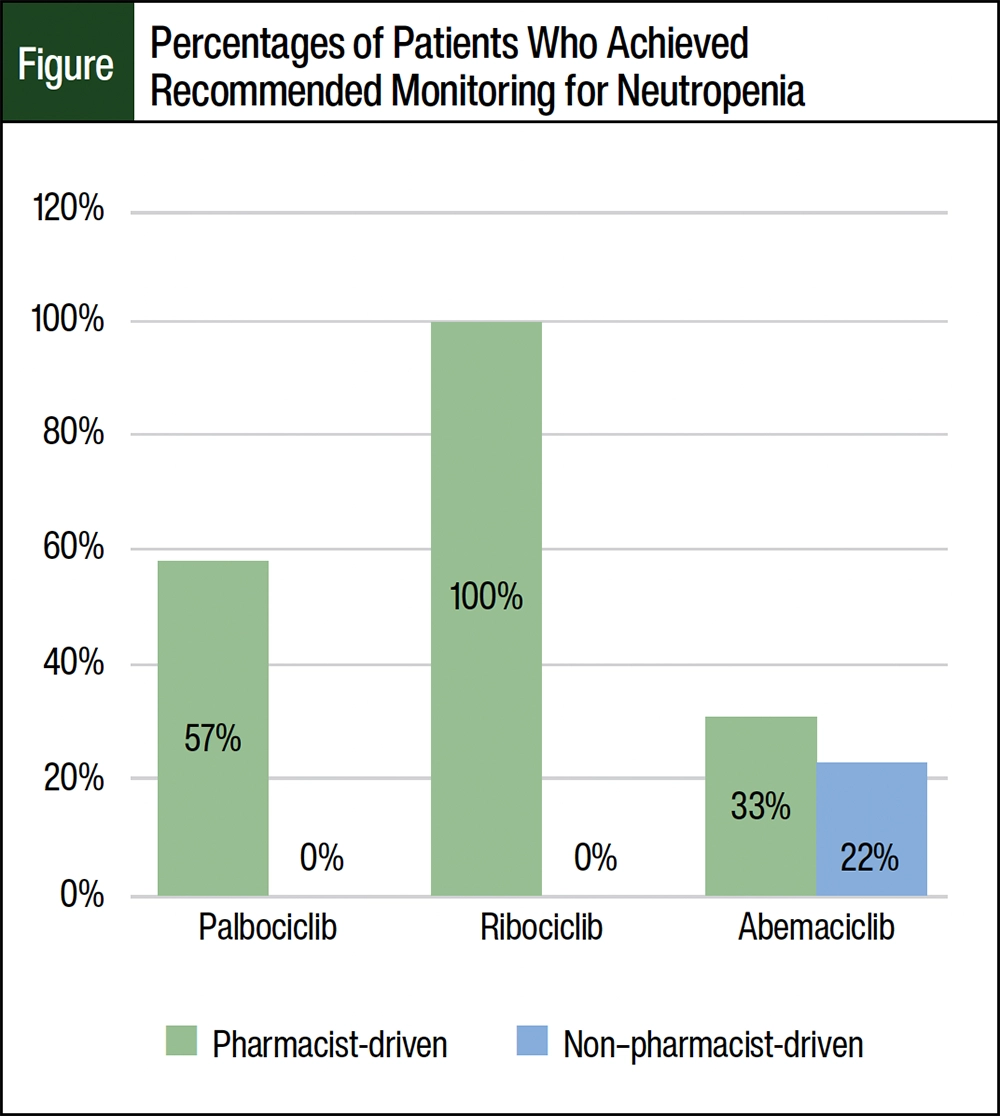
Overall, 63% of the patients managed by oncology clinical pharmacists in the MIP achieved the manufacturer-recommended laboratory monitoring for neutropenia, which is outlined in Appendix I. The percentages of patients receiving CDK4/6 inhibitors was 57%, 100%, and 33% in the palbociclib, ribociclib, and abemaciclib groups, respectively (Figure). It is important to note that only 1 patient was receiving ribociclib while being managed by an oncology clinical pharmacist in an MIP. None of the patients in the palbociclib and ribociclib groups who were receiving CDK4/6 inhibitors from external fulfillment achieved the recommended laboratory monitoring. Approximately 22% of the patients receiving abemaciclib achieved the recommended laboratory monitoring. The percentage of patients who achieved the manufacturer-recommended laboratory monitoring for neutropenia are shown in the Figure.
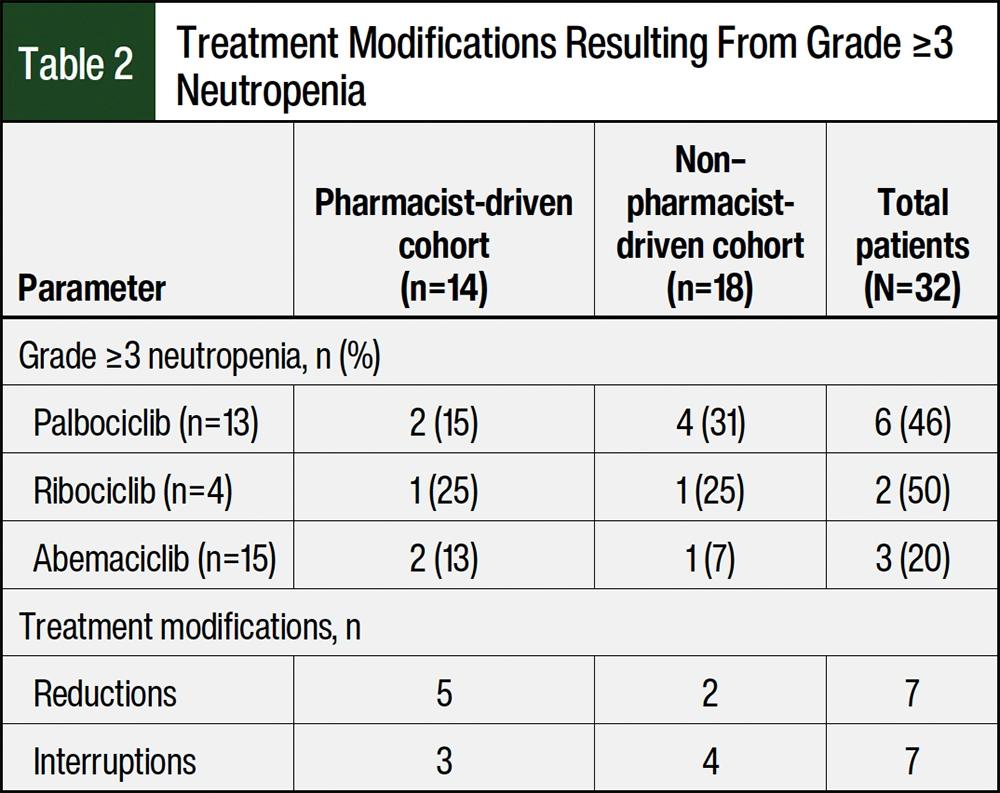
More than 20% of the patients in both groups had grade ≥3 neutropenia when examining all 3 CDK4/6 inhibitors, including patients who required a dose reduction or treatment interruption (Table 2). For patients who were managed by oncology clinical pharmacists, the incidences of grade ≥3 neutropenia were 15% and 13% in the patients receiving palbociclib and abemaciclib, respectively. In the patients managed through external fulfillment, grade ≥3 neutropenia occurred in 31% and 7% of the patients receiving palbociclib and abemaciclib, respectively. In both groups, the overall incidences of grade ≥3 neutropenia in the patients who received ribociclib were 25% (Table 2).
A total of 5 (27.8%) patients had grade 3 neutropenia and 1 (6%) patient had grade 4 neutropenia in the external fulfillment group. Only 1 patient with grade 3 neutropenia had a 1-week treatment delay, whereas 4 of the patients had a dose reduction or treatment delay of ≥14 days. In 2 of these cases, the patients had a dose reduction after prolonged grade 3 neutropenia in addition to a treatment delay. The fifth patient with grade 3 neutropenia did not have appropriate interventions, despite having an ANC of >1000 cells/µL for 2 cycles. The patient with grade 4 neutropenia had a treatment delay of 20 days after missing timely CBC orders during cycle 2. Her ANC was 350 cells/µL and remained approximately 500 cells/µL, leading to a delayed start of the next cycle. The patient eventually restarted treatment at a lower dose of abemaciclib after 20 days.
A total of 5 (35.7%) patients had grade 3 neutropenia while being managed by oncology clinical pharmacists, and none of the patients had grade 4 neutropenia. In all 5 cases, the oncology clinical pharmacists identified the neutropenia during cycles 1 or 2 and followed the manufacturer-recommended interventions. For example, a patient receiving palbociclib had an ANC of 530 cells/µL on cycle 1 day 15, so the oncology clinical pharmacist made a recommendation to repeat the CBC testing on day 22 per palbociclib’s prescribing information.
Discussion
This prospective, single-center study was performed to evaluate the outcomes of a pharmacist-driven approach in monitoring for CDK4/6 inhibitor hematologic adverse events in women with advanced breast cancer. The results reveal that patients receiving CDK4/6 inhibitors who are managed by oncology clinical pharmacists were more likely than patients who received their medication through external fulfillment to achieve manufacturer-recommended laboratory monitoring for neutropenia. The higher incidence of grade ≥3 neutropenia that was observed in the group managed by oncology clinical pharmacists was likely a result of the close monitoring the patients received, especially during cycles 1 and 2 of treatment. Grade ≥3 neutropenia was managed appropriately early in therapy, which prevented prolonged treatment interruptions or changes in therapy. The average delayed treatment days were 23 days in the external fulfillment cohort and 16 days in the pharmacist-driven cohort, demonstrating the significance of pharmacist-led interventions in oral oncolytic therapy. It is important to note that patients in the pharmacist-driven group who did not achieve the recommended laboratory testing only missed 1 CBC order, typically during cycle 3 or 4.
The patients receiving CDK4/6 inhibitors in the external fulfillment cohort were more likely than the patients managed by oncology clinical pharmacists to miss ≥3 CBC orders. Each patient was expected to complete 8 CBC orders during the first 6 treatment cycles per the manufacturer’s recommendations, equating to a total of 144 CBC orders in the external fulfillment cohort and 112 CBC orders in the pharmacist-driven cohort. In the pharmacist-driven cohort, there were only 20 CBC orders that were missed while receiving treatment. However, there were 54 CBC orders that were missed while receiving treatment in the external fulfillment cohort. In 3 patients, the CBC orders were performed only during cycle 1 of treatment, leading to grade ≥3 neutropenia that was not immediately addressed. The 2 patients whose ANC remained at <1000 cells/µL highlight the role of oncology clinical pharmacists in an interdisciplinary team. In both cases, close monitoring by oncology clinical pharmacists could have led to the appropriate management of hematologic adverse events in the early treatment cycles.
It is interesting to note that 9 patients who were receiving abemaciclib had diarrhea of any grade, and 3 of those events were grade ≥2. One event of diarrhea and 1 event of severe chest pain led to a dose reduction of abemaciclib. Another general observation was the inconsistent baseline evaluation of QTc intervals in patients receiving ribociclib. These adverse events were documented by oncology clinical pharmacists and were discussed with providers during the biweekly check-ins that are part of the routine follow-up process for all patients receiving oral oncolytic agents from the MIP. The pharmacists work in close proximity with providers for collaborative dispensing and clinical care. The inconsistent baseline evaluation of QTc intervals highlighted another opportunity to expand clinical practice, and standardized monitoring by oncology clinical pharmacists was implemented as a result of this project.
Previous studies have reported neutropenia as the most frequent adverse event with CDK4/6 inhibitors.7-9 In the PALOMA-3 trial, neutropenia of any grade occurred in 81% of the patients in the palbociclib plus fulvestrant arm, and 65% of those neutropenia events were grade 3 or 4.7 Febrile neutropenia was uncommon.7 In the MONALEESA-7 trial, the most common (61%) grade 3 or 4 adverse event that led to dose reduction was neutropenia in patients receiving ribociclib, plus tamoxifen or a nonsteroidal aromatase inhibitor, and goserelin, and the median time to the first dose reduction was approximately 3 months, demonstrating the importance of close monitoring, especially at the beginning of treatment.8 In the MONARCH-3 trial, any-grade neutropenia occurred in 43.7% of patients receiving abemaciclib plus aromatase inhibitor, of which 23.9% of those events were grade 3 or 4. Only 1 patient in the treatment arm at the interim analysis had febrile neutropenia.9 Our findings reveal similar observations to these studies. The manufacturers for each CDK4/6 inhibitor have recommended laboratory monitoring for clinicians to ensure that patients are appropriately monitored. However, the monitoring for neutropenia with CDK4/6 inhibitors may vary per institution, and oncology clinical pharmacists working at St Luke’s Cancer Institute did not have specific documentation for these agents before this study.
Oncology clinical pharmacists working in the MIP have an opportunity to expand clinical services and improve the monitoring for neutropenia under their CPA. Before this study, pharmacists documented the most recent date of CBC order before dispensing a CDK4/6 inhibitor. Findings from this study led to the expansion of clinical documentation within the medical records (Appendix II), which includes the neutrophil count value and other pertinent laboratory values with CDK4/6 inhibitors. Although laboratory orders for monitoring neutropenia are built into the electronic medical record for each CDK4/6 inhibitor, there are numerous opportunities to ensure that all patients achieve the recommended monitoring, regardless of the fulfillment location. Oncology clinical pharmacists completed thorough clinical reviews and adhered to the appropriate laboratory testing during the first 4 months of treatment. The aim of expanding the clinical documentation is to address the missed laboratory orders in a timely manner and to ensure that pharmacists can order CBCs under the CPA, thus improving the quality of patient care in the institution. In addition, the findings were reported to leadership in support for additional pharmacist resources in the MIP to provide high-quality patient care.
Limitations
A limitation of this study was the difficulty in finding information on patients who received CDK4/6 inhibitors through external fulfillment mechanisms. Treatment delays or interruptions were not clearly noted, and a thorough investigation of the chart review had to be performed to ensure accuracy.
Another limitation is that 4 patients (2 patients in each of the cohorts) were lost during the study because they initiated a clinical trial, they transitioned their care to another cancer center, or they had a change in therapy. These patients were included in the study even though they did not complete ≥6 treatment cycles.
The strengths of this study include a similar incidence of hematologic adverse events with CDK4/6 inhibitors compared with the current literature. It also reveals the importance of educating oncology clinical pharmacists on timely CBC orders. Oncology clinical pharmacists were more likely than clinicians overseeing external fulfillment to adhere to appropriate laboratory testing during the first 4 months of treatment.
Pharmacists play a pivotal role in the timely monitoring of hematologic adverse events with CDK4/6 inhibitors to prevent dosage modifications and treatment delays. Performing routine clinical evaluations of and counseling for adverse-event management helps improve outcomes for patients with cancer. Future directions include extending the duration of prospective data analysis to evaluate the impact of the expansion in clinical documentation within the medical records and evaluating the incidence of other adverse events, such as cardiac or gastrointestinal adverse events.
Conclusion
A pharmacist-driven approach to monitoring for neutropenia with CDK4/6 inhibitors in women with advanced breast cancer results in improved adherence to the manufacturers’ recommendations. More important, this article shows the positive downstream effects of diligent monitoring for adverse events and continuity of care, including decreased grade 3/4 adverse events and shortened treatment delays, thus leading to better patient outcomes. Oncology clinical pharmacists specializing in oral oncolytic treatments have many opportunities to implement similar pharmacist-driven approaches to the management of adverse events for other medications to improve patients’ clinical outcomes.
Acknowledgments
The authors would like to acknowledge Stephanie Matta, PharmD, BCOP, Quincy Ostrem, PharmD, MBA, and Bryce Benson, PharmD, who contributed to the study design.
Author Disclosure Statement
Dr De Raya and Dr Modlin have no conflicts of interest to report.
References
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. Erratum in: CA Cancer J Clin. 2020;70:313.
- Breast Cancer Facts & Figures 2022-2024. American Cancer Society. Accessed June 14, 2023. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/2022-2024-breast-cancer-fact-figures-acs.pdf
- Shah M, Nunes MR, Stearns V. CDK4/6 inhibitors: game changers in the management of hormone receptor-positive advanced breast cancer? Oncology (Williston Park). 2018;32:216-222.
- Ibrance (palbociclib) tablets, for oral use [prescribing information]. Pfizer; September 2023. Accessed March 27, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/212436s005s006lbl.pdf
- Kisqali (ribociclib) tablets, for oral use [prescribing information]. Novartis; August 2023. Accessed March 27, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/209092s016lbl.pdf
- Verzenio (abemaciclib) tablets, for oral use [prescribing information]. Eli Lilly; March 2023. Accessed March 27, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/208716s010s011lbl.pdf
- Cristofanilli M, Turner NC, Bondarenko I, et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2016;17:425-439. Errata in: Lancet Oncol. 2016;17:e136; Lancet Oncol. 2016;17:e270.
- Tripathy D, Im SA, Colleoni M, et al. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): a randomised phase 3 trial. Lancet Oncol. 2018;19:904-915.
- Johnston S, Martin M, Di Leo A, et al. MONARCH 3 final PFS: a randomized study of abemaciclib as initial therapy for advanced breast cancer. npj Breast Cancer. 2019;5:5.
- Lynce F, Shajahan-Haq AN, Swain SM. CDK4/6 inhibitors in breast cancer therapy: current practice and future opportunities. Pharmacol Ther. 2018;191:65-73.
- Taraba J, Golbach A, Smith M, et al. Development and implementation of a pharmacist-led virtual clinic improve the management of patients with metastatic breast cancer receiving CDK4/6 inhibitors. J Hematol Oncol Pharm. 2022;12:14. Abstract CR04.