Connecting Science to Practice
Cytokine release syndrome (CRS) is a systemic inflammatory response
caused by the release of inflammatory cytokines, and it is a
common and potentially fatal complication after CAR T-cell therapy.
Tocilizumab, an interleukin-6 receptor antagonist, can be
given with dexamethasone as first-line treatment for CRS. The role
of other cytokine inhibitors is not well-established in this application,
but alternative treatments have been used for refractory CRS.
This single-center, retrospective study assessed if anakinra can
minimize the duration of CRS after initial therapy with tocilizumab.
In this study, patients who received anakinra after an initial
dose of tocilizumab and patients who received multiple doses of
tocilizumab had similar durations of CRS, suggesting that anakinra
may be an acceptable alternative therapy for the treatment of CRS
in the relapsed or refractory setting.
Cytokine release syndrome (CRS) is characterized as a systemic inflammatory response caused by the release of inflammatory cytokines, such as interleukin (IL)-6, interferon-γ, tumor necrosis factor-α, IL-10, and IL-2, by activated lymphocytes and myeloid cells.1,2 IL-1, IL-5, IL-8, granulocyte-macrophage colony-stimulating factor, and serum biochemical markers, including C-reactive protein and ferritin, are also elevated in patients with CRS.1,2 These cytokines play a key role in the pathophysiology and clinical manifestations of CRS, which include fever, hypotension, and organ dysfunction.3
In 2019, the American Society for Transplantation and Cellular Therapy (ASTCT) published consensus diagnostic criteria1 that have been adapted by many institutions that administer CAR T-cell therapy. In 2020, the Society for Immunotherapy of Cancer (SITC) established an expert panel to provide guidance on managing immune effector cell–related adverse events (AEs).4 Based on the SITC’s practice guidelines, tocilizumab (an IL-6 receptor antagonist) is recommended for the treatment of grade 2 CRS and may be given, in combination with a corticosteroid such as dexamethasone, every 8 hours for up to 4 doses as first-line treatment.4,5 The role of other cytokine inhibitors is not well-established in this setting, but therapy alternatives such as anakinra and siltuximab have been used for the treatment of refractory CRS in adults and pediatric populations.4,6
Recently, the national supply of tocilizumab has diminished because of its role of improving the outcomes of patients with COVID-19.7 Alternative drugs are now being investigated for the treatment of CRS. Anakinra, an IL-1 receptor antagonist, is currently approved by the FDA for the treatment of rheumatoid arthritis and neonatal-onset multisystem inflammatory disease.8 Anakinra is also used off-label for the treatment of secondary hemophagocytic lymphohistiocytosis, another AE that may be associated with CAR T-cell therapy.9-12 Here, we explore the role of anakinra for the management of CRS.
Currently, there are limited data regarding the use of anakinra for the treatment of CRS. In 2 animal model studies, IL-1 inhibition prevented CRS and immune effector cell–associated neurotoxicity syndrome (ICANS).13,14 A recent case series by MD Anderson Cancer Center reported a clinical benefit in 4 of 8 patients who received treatment with anakinra to mitigate axicabtagene ciloleucel–associated AEs.15 However, the indication for anakinra was high-grade ICANS in 6 patients and hemophagocytic lymphohistiocytosis in 2 patients.15 Clinical trials that have investigated the prophylactic use of anakinra for CRS have been recently completed or are ongoing (NCT04432506,16 NCT04359784,17 and NCT0414843018), but to the best of our knowledge, there are no published study data that have evaluated the use of anakinra as a treatment for CRS.
During the study period at our institution, the patients received tocilizumab 8 mg/kg intravenously (IV) and dexamethasone 10 mg IV once for the initial treatment of grade 2 CRS (Appendix Table A1). Before the COVID-19 pandemic, patients with recurrent and/or refractory CRS were receiving additional doses of tocilizumab. In April 2020, the use of anakinra was added to our institutional CRS management guidelines in an effort to conserve tocilizumab during national shortages.19 Based on these updated guidelines, patients with recurrent or refractory grade 2 CRS after the initial dose of tocilizumab were receiving anakinra and dexamethasone, and patients with grade 3 or 4 CRS were receiving all 3 agents concurrently.
The objective of this study was to assess if anakinra is a feasible treatment option to minimize the duration of CRS after initial management with tocilizumab.
Methods
This single-center, retrospective study included adults aged ≥18 years who had CRS after receiving CAR T-cell therapy at Stanford Health Care from January 1, 2019, to September 30, 2021. Based on our institutional guidelines, patients with grade 2 CRS were initially receiving a dose of tocilizumab 8 mg/kg IV (maximum, 800 mg) and dexamethasone 10 mg IV. Patients with recurrent and/or refractory grade 2 CRS received anakinra 100 mg subcutaneously every 6 hours for 3 days concurrently with dexamethasone 10 mg IV every 6 hours. Patients with grade 3 CRS received tocilizumab, anakinra, and dexamethasone at the previously mentioned doses, and patients with grade 4 CRS received tocilizumab, anakinra, and methylprednisolone instead of dexamethasone (Appendix Table A1). The dose of anakinra was based on the data that were available at the time for the treatment of macrophage activation syndrome–associated hemophagocytic lymphohistiocytosis, which overall suggested that higher doses of 100 to 200 mg 3 to 4 times daily were needed, especially in patients with refractory disease.20-22 The short half-life of anakinra (4-6 hours)8 may justify more frequent dosing to rapidly achieve therapeutic IL-1 inhibition.
The patients who received anakinra after the initial dose of tocilizumab (ie, the anakinra group) were compared with the patients who received at least 2 doses of tocilizumab under the standard management strategy (ie, the tocilizumab group). At least 2 doses of tocilizumab were required so that the tocilizumab treatment group would be reflective of a population with refractory or recurrent CRS. The patients in the anakinra group were permitted to receive additional doses of tocilizumab for the treatment of ongoing CRS after starting therapy with anakinra, and treatment with anakinra could be continued for longer than 3 days at the discretion of the provider. Patients were excluded from the study if they received anakinra or tocilizumab for other indications, including ICANS without concurrent CRS. The data were collected until the resolution of CRS and ICANS or until day 100, whichever occurred first.
All of the patients received infection prophylaxis as the standard of care, including acyclovir for antiviral prophylaxis and atovaquone for Pneumocystis jirovecii pneumonia prophylaxis. Treatment with both agents was continued for 18 to 24 months, and atovaquone could be subsequently transitioned to trimethoprim plus sulfamethoxazole for patients with adequate neutrophil recovery. No routine bacterial prophylaxis was used. Antimold prophylaxis was considered when patients received >4 days of treatment with high-dose intravenous steroids.
CRS was diagnosed, graded, and managed according to Stanford Health Care’s CRS guidelines (Appendix Table A1), which followed the 2019 ASTCT consensus grading system.1 The maximum grade documented by the provider was used to assess the severity of CRS. CRS resolution was defined as being afebrile and hemodynamically stable (eg, not receiving vasopressors) for 24 hours. The recurrence of CRS was defined as symptoms reoccurring after full resolution of CRS and not receiving treatment for 24 hours. The total corticosteroid dose was defined as the total dosage of corticosteroids administered during the hospital admission, which was calculated in dexamethasone equivalents.
ICANS was graded and managed according to Stanford Health Care’s ICANS guidelines (Appendix Table A2). ICANS resolution was defined as the return to the patient’s baseline neurologic function and the resolution of symptoms as documented by clinical assessments.
The data on survival and response to CAR T-cell therapy were obtained from the centralized Stanford Blood and Marrow Transplant and Cancer Cell Therapy database. These assessments were completed by Stanford Blood and Marrow Transplant providers per Cancer Cell Therapy protocols.
The primary outcome of the study was the duration of CRS. The secondary outcomes included the onset of CRS, which was defined as the time from CAR T-cell infusion to a diagnosis of CRS; maximum grade of CRS; time to resolution of CRS; recurrence of CRS; peak C-reactive protein (measured from start of lymphodepletion to discharge); time to the start of tocilizumab treatment after CAR T-cell infusion; total doses of tocilizumab received; time to the start of anakinra treatment after CAR T-cell infusion; duration of anakinra treatment; cumulative corticosteroid dose; incidence of ICANS; onset of ICANS (defined as the time from CAR T-cell infusion to ICANS diagnosis); duration of ICANS; maximum grade of ICANS; incidence and type of infection (from the start of lymphodepletion to discharge); 30-day and 100-day overall survival; and the overall response rate (ORR; partial response or better) to CAR T-cell therapy at 30 days and 90 days.
Statistical Analysis
The baseline characteristics were described with absolute frequencies or medians. The categorical variables were compared by the chi-square test. The continuous variables were compared by the Mann–Whitney U test. Statistical significance was considered if P<.05.
Results
A total of 79 patients were screened, including 27 patients who received anakinra and 52 patients who received tocilizumab after CAR T-cell therapy for the management of CRS during the study period. After applying the exclusion criteria, 30 total patients were included in the study with 8 patients in the anakinra group and 22 patients in the tocilizumab group (Table 1). The most common reason for exclusion from the anakinra group was receiving anakinra for ICANS without concurrent CRS. The median age was 69 years (range, 47-83 years) and 66 years (range, 31-82 years) in the anakinra and tocilizumab groups, respectively (P=.78). Overall, the baseline characteristics were balanced between the groups (Table 1).
The median duration of CRS was 6 days (range, 3-9 days) in the anakinra group and 7 days (range, 2-21 days) in the tocilizumab group (P=.45; Table 2).
The CRS-related secondary outcomes were similar between the groups (Table 2). The onset of CRS occurred at a median of 1 day (range, 1-5) and 3 days (range, 0-8) after CAR T-cell infusion in the anakinra and tocilizumab groups, respectively (P=.45). Most patients had low-grade CRS (grade 1, 0% with anakinra vs 9.1% with tocilizumab, P=.38; grade 2, 88% vs 82%, respectively, P=.71). In all, 1 of 8 (12.5%) patients in the anakinra group and 2 of 22 (9.1%) patients in the tocilizumab group had grade ≥3 CRS (P=.78). The median peak C-reactive protein was 11 mg/dL (range, 0.7-18) in the anakinra group and 7.3 mg/dL (range, 1.2-28) in the tocilizumab group (P=.62). CRS resolved in all of the patients in the anakinra group and in 86% of the patients in the tocilizumab group within 24 hours after receiving CRS treatment (P=.27). CRS did not recur in the anakinra group and recurred in 2 (9.1%) patients in the tocilizumab group (P=.38). Tocilizumab was started in a median of 3 days (range, 1-6) after CAR T-cell infusion in the anakinra group and in a median of 5 days (range, 0-10) in the tocilizumab group (P=.22).
As expected, treatment with tocilizumab was lower in the anakinra group than in the tocilizumab group. The median number of tocilizumab doses was 1 (range, 1-4) in the anakinra group compared with 2 (range, 2-4) in the tocilizumab group (P=.02). One patient in each group received 4 doses of tocilizumab for grade 3 CRS. Anakinra was started at a median of 4 days (range, 1-7 days) after CAR T-cell infusion and was received for a median duration of 4 days (range, 3-7 days). The median total equivalent dose of dexamethasone in the anakinra group was 253 mg (range, 50-1555 mg) and 117 mg (range, 10-729 mg) in the tocilizumab group (P=.07).
ICANS occurred in 4 (50%) patients in the anakinra group and in 15 (68%) patients in the tocilizumab group (P=.36; Table 2). The onset of ICANS occurred at a median of 3 (range, 0-6 days) days and 5 (range, 0-10 days) days after CAR T-cell infusion in the anakinra and tocilizumab groups, respectively (P=.27). The median duration of ICANS was 5 days (range, 2-7 days) in the anakinra group and 4 days (range, 2-39 days) in the tocilizumab group (P=.36). The rates of grade ≥3 ICANS were similar in both groups (38% vs 32%, respectively; P=.77).
Infections occurred in 13% and 23% of patients in the anakinra and tocilizumab groups, respectively (P=.54; Table 2). Most cases of infection were bacterial, including 1 patient in the anakinra group and 3 patients in the tocilizumab group (P=.46). A total of 2 (9%) patients in the tocilizumab group had a viral infection (P=.78). None of the patients had a fungal infection.
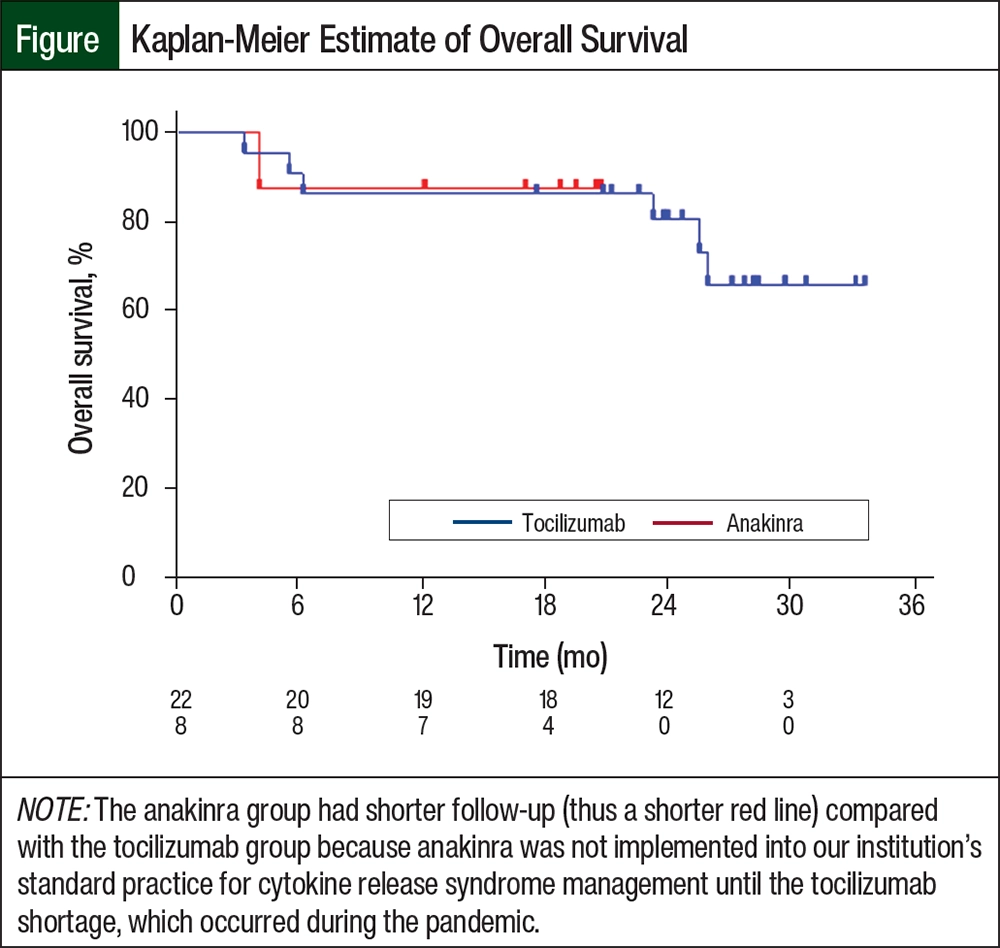
The 30-day and 100-day overall survival rates were similar between the groups (Table 2, Figure). There were no deaths before day 30 in either group. By day 100, 1 of 22 (4.5%) patients in the tocilizumab group died as a result of severe deconditioning that was secondary to ICANS at a maximum of grade 3 that ultimately resolved to grade 0, but with waxing and waning delirium that ultimately culminated in a decision to proceed to hospice. None of the patients in the anakinra group died before day 100 (P=.54).
The 30-day and 90-day ORRs were also similar between the groups (Table 2). By day 30, 88% of the patients in the anakinra group and all of the patients in the tocilizumab group had a partial response or better to CAR T-cell therapy (P=.09). By day 90, 3 of 5 (60%) patients in the anakinra group with evaluable data had a partial response or better, and 12 of 18 (67%) evaluable patients in the tocilizumab group had a partial response or better (P=.41). Of note, the 90-day response data were not available for 3 patients in the anakinra group and for 4 patients in the tocilizumab group.
Discussion
In this study, the median duration of CRS was similar between the patients who received anakinra after an initial dose of tocilizumab and those who received repeat doses of tocilizumab. The median durations of CRS in both groups were consistent with those in clinical trials of axicabtagene ciloleucel,23,24 which was the most frequently received CAR T-cell therapy in our study. More important, the patients in the anakinra group received significantly fewer tocilizumab doses while achieving similar CRS-related outcomes to the patients who did not receive anakinra. Notably, most patients in the anakinra group did not require additional doses of tocilizumab, and the CRS resolved and did not recur in any of the patients. Thus, this result shows that there is a benefit to treatment with anakinra and that there is a potential role for IL-1 inhibition in patients with refractory or recurrent CRS.
Interestingly, the total corticosteroid dose was higher in the anakinra group than in the tocilizumab group. Although this difference did not reach statistical significance, it contrasts a recent case series that demonstrated the clinical efficacy of anakinra as a potential steroid-sparing agent in the treatment of CAR T-cell therapy–related AEs, especially in patients with ICANS.15 It should be noted that the retrospective nature of our study limited our ability to differentiate the corticosteroid use in the setting of overlapping AEs. Given that at least 50% of the patients in both groups had CRS with concurrent ICANS, definitive conclusions cannot be drawn regarding the role of anakinra as a steroid-sparing agent.
The use of anakinra did not significantly affect the overall incidence of infection, which was low in both groups. Although this is somewhat reassuring because treatment with anakinra and treatment with tocilizumab are associated with an increased risk for infections,5,8 our study is not truly powered to evaluate this potential complication.
More important, it is encouraging that both groups in this study had similar survival and CAR T-cell therapy response rates. Given the concern that steroids may hinder antitumor response,25 this finding supports further investigation of anakinra as a steroid-sparing agent for the treatment of CAR T-cell therapy–related AEs.
Recently, Gazeau and colleagues published a retrospective, multicenter study of 43 patients that evaluated the safety of anakinra for the treatment of refractory CRS and ICANS after CAR T-cell therapy.26 In this study, the doses of anakinra ranged from 100 to 200 mg daily subcutaneously or IV (low dose) to >200 mg daily IV (high dose), and doses up to 12 mg/kg daily IV were safe and feasible. The median time to CRS and ICANS resolution after starting anakinra was 7 days among patients who received high-dose anakinra, and the high ORR of 77% to CAR T-cell therapy suggested that anakinra did not compromise the efficacy of CAR T-cell therapy.26 These results are consistent with the findings of our study.
Limitations
Our study has several limitations. Our study’s retrospective design and small sample size may have led to selection bias by limiting our ability to control for confounding variables, such as differences in tumor histology, tumor burden, and the CAR T-cell therapy received. Although standardized guidelines were used by all treating providers, variations in CRS and ICANS grading and differences in management between providers cannot be excluded. These initial results support the need for a larger, well-powered study to confirm anakinra’s role as a sufficient treatment for refractory CRS.
Conclusion
In this study, patients who received anakinra after an initial dose of tocilizumab had similar outcomes to patients who received multiple doses of tocilizumab. This outcome shows that anakinra may be an acceptable alternative to tocilizumab for the treatment of CRS that results from CAR T-cell therapy in the relapsed or refractory disease setting. Given intermittent national shortages of tocilizumab, further studies that evaluate the use of anakinra for the treatment of refractory CRS are warranted.
Author Disclosure Statement
Dr Patel is an employee of Amgen; Ms Latchford is on the Speakers Bureau at Kite-Gilead and Bristol Myers Squibb; Dr Frank is on the Advisory Board of Kite-Gilead, on the Advisor Board and received research support from Cargo and Adaptive Biotechnologies, and received research support from Allogene Therapeutics. Dr Kim and Dr Domingo have no conflicts of interest to report.
References
- Lee DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625-638.
- Wang Z, Han W. Biomarkers of cytokine release syndrome and neurotoxicity related to CAR-T cell therapy. Biomark Res. 2018;6:4.
- Riegler LL, Jones GP, Lee DW. Current approaches in the grading and management of cytokine release syndrome after chimeric antigen receptor T-cell therapy. Ther Clin Risk Manag. 2019;15:323-335.
- Maus MV, Alexander S, Bishop MR, et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune effector cell-related adverse events. J Immunother Cancer. 2020;8:e001511.
- Actemra (tocilizumab) injection, for intravenous use [prescribing information]. Genentech; September 2024. Accessed October 15, 2024. www.gene.com/download/pdf/actemra_prescribing.pdf
- Lee DW, Gardner R, Porter DL, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124:188-195. Errata in: Blood. 2015;126:1048; Blood. 2016;128:1533.
- Morris EC, Neelapu SS, Giavridis T, Sadelain M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol. 2022;22:85-96.
- Kineret (anakinra) injection, for subcutaneous use [prescribing information]. Sobi; December 2020. Accessed October 15, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2020/103950s5189lbl.pdf
- Rajasekaran S, Kruse K, Kovey K, et al. Therapeutic role of anakinra, an interleukin-1 receptor antagonist, in the management of secondary hemophagocytic lymphohistiocytosis/sepsis/multiple organ dysfunction/macrophage activating syndrome in critically ill children. Pediatr Crit Care Med. 2014;15:401-408.
- Shakoory B, Carcillo JA, Chatham WW, et al. Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: reanalysis of a prior phase III trial. Crit Care Med. 2016; 44:275-281.
- Hines MR, Keenan C, Maron Alfaro G, et al. Hemophagocytic lymphohistiocytosis-like toxicity (carHLH) after CD19-specific CAR T-cell therapy. Br J Haematol. 2021;194:701-707.
- Lichtenstein DA, Schischlik F, Shao L, et al. Characterization of HLH-like manifestations as a CRS variant in patients receiving CD22 CAR T cells. Blood. 2021;138:2469-2484.
- Norelli M, Camisa B, Barbiera G, et al. Monocyte-derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nat Med. 2018;24:739-748.
- Giavridis T, van der Stegen SJC, Eyquem J, et al. CAR T cell-induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade. Nat Med. 2018;24:731-738.
- Strati P, Ahmed S, Kebriaei P, et al. Clinical efficacy of anakinra to mitigate CAR T-cell therapy–associated toxicity in large B-cell lymphoma. Blood Adv. 2020;4:3123-3127.
- Anakinra for the reduction of CAR-T toxicity in patients with relapsed or refractory large B-cell lymphoma. NLM identifier: NCT04432506. Updated May 31, 2024. Accessed October 16, 2024. https://clinicaltrials.gov/study/NCT04432506?tab=history&a=13
- Anakinra for the prevention of cytokine release syndrome and neurotoxicity in patients with B-cell non-Hodgkin lymphoma receiving CD19-targeted CAR-T cell therapy. NLM identifier: NCT04359784. Updated July 18, 2024. Accessed October 16, 2024.
- A study of anakinra to prevent or treat severe side effects for patients receiving CAR-T cell therapy. NLM identifier: NCT04148430. Updated February 26, 2024. Accessed October 16, 2024.
- American Society of Health-System Pharmacists. Current drug shortages: tocilizumab injection. July 29, 2022. Accessed August 8, 2024. www.ashp.org/drug-shortages/current-shortages/drug-shortage-detail.aspx?id=744#
- Kahn PJ, Cron RQ. Higher-dose anakinra is effective in a case of medically refractory macrophage activation syndrome. J Rheumatol. 2013;40:743-744.
- Wohlfarth P, Agis H, Gualdoni GA, et al. Interleukin 1 receptor antagonist anakinra, intravenous immunoglobulin, and corticosteroids in the management of critically ill adult patients with hemophagocytic lymphohistiocytosis. J Intensive Care Med. 2019;34:723-731.
- Mehta P, Cron RQ, Hartwell J, et al. Silencing the cytokine storm: the use of intravenous anakinra in haemophagocytic lymphohistiocytosis or macrophage activation syndrome. Lancet Rheumatol. 2020;2:e358-e367.
- Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377:2531-2544.
- Locke FL, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel as second-line therapy for large B-cell lymphoma. N Engl J Med. 2022;386:640-654.
- Maude SL, Barrett D, Teachey DT, Grupp SA. Managing cytokine release syndrome associated with novel T cell-engaging therapies. Cancer J. 2014;20:119-122.
- Gazeau N, Liang EC, Wu QV, et al. Anakinra for refractory cytokine release syndrome or immune effector cell-associated neurotoxicity syndrome after chimeric antigen receptor T cell therapy. Transplant Cell Ther. 2023;29:430-437.