Granulocyte colony-stimulating factor (G-CSF) is often used after a hematopoietic stem-cell transplant (HSCT) to expedite neutrophil recovery after high-dose chemotherapy. Prolonged neutropenia after HSCT increases the risk for infectious complications and can contribute significantly to morbidity and mortality.1-3 Several studies have shown that G-CSF use can increase neutrophil recovery by 1 to 6 days compared with placebo in autologous and in allogeneic HSCT.4-8 In addition, studies have shown that G-CSF use can lead to cost-savings, by decreasing posttransplant length of hospital stay, although no significant differences in time to platelet engraftment and days of febrile neutropenia were found.4,5,9,10
Unlike autologous transplants, in which consensus has been reached regarding G-CSF use, several concerns exist regarding the use of G-CSF in allogeneic HSCT. The American Society of Clinical Oncology (ASCO) 2015 guideline update on the use of white blood cells growth factors includes strong recommendations for the use of G-CSF to accelerate neutrophil recovery after an autologous HSCT; this recent version was updated to include weak recommendations for use after an allogeneic HSCT.11 G-CSF use after allogeneic transplantation has been linked to an increase in acute graft-versus-host disease (GVHD) and treatment-related mortality.12-15 The update was based on a meta-analysis that concluded that G-CSF use after allogeneic transplants reduced the risk for documented infections and did not have a statistically significant effect on acute GVHD or on treatment-related mortality.16
G-CSF can be administered safely in patients as an intravenous (IV) infusion or subcutaneous (SQ) injection. Although the preferred route of administration is SQ, the IV route of administration is often used at our institution in patients undergoing HSCT because of its ease of administration, patient convenience, and reduced bruising at the injection site in patients with thrombocytopenia.11
Pharmacokinetic analyses in healthy adults showed G-CSF absorption to be a composite of zero-order and first-order processes, and its clearance governed by the binding of filgrastim to G-CSF receptors.17,18 In both IV and SQ delivery methods, G-CSF rapidly binds to its receptor and is internalized, which activates signal transduction and transcription factors, ultimately leading to a rapid increase in the number of serum neutrophils from the marrow storage pool.17,18
The primary difference between SQ and IV administration is based on how quickly this process occurs; with IV administration, maximum G-CSF concentrations are achieved almost immediately after infusion. SQ G-CSF has to be absorbed systemically from the tissue and peak concentrations are not seen for several hours.18 A previous study has shown earlier peaks of neutrophil counts with IV administration but larger increases in neutrophil counts with SQ administration.17
At our institution (Methodist University Hospital in Memphis), G-CSF (ie, filgrastim) is used in patients undergoing either autologous or allogeneic HSCT to enhance neutrophil recovery. The route of administration was recently changed from IV to SQ to facilitate the opportunity to evaluate outcomes based on the different routes of administration.
The purpose of this study was to compare the time to neutrophil engraftment in HSCT patients receiving SQ and IV G-CSF. The secondary outcome measures were to compare time to neutrophil engraftment between autologous and allogeneic HSCT, incidence of febrile neutropenia, occurrence of documented infection, in-hospital all-cause mortality, and length of stay.
Methods
This study was conducted as a single-center, retrospective chart review of patients admitted to our institution between July 1, 2015, and December 15, 2016, who underwent HSCT and received G-CSF. Patients were identified via an internal stem-cell transplant patient database and the institution’s electronic medical record. Patients were included if they were older than 18 years, underwent either allogeneic or autologous HSCT, and received G-CSF posttransplant for a minimum of 7 doses. Patients were excluded if they missed 2 or more consecutive doses of G-CSF. Between July 2015 and April 2016, G-CSF was administered at our institution using mainly the IV route, and this practice was changed in April 2016, so that the majority of patients were switched to the SQ administration based on provider preference.
The primary study outcome was to determine if there was a difference in days to neutrophil engraftment in patients undergoing HSCT who received SQ versus IV administration of G-CSF. Neutrophil engraftment was measured from the day of transplant (day 0) to the first day of absolute neutrophil count (ANC) ≥500 cells/mcL for 3 consecutive days. Secondary outcomes included days to neutrophil recovery, defined as the number of days from day 1 of G-CSF to the first day of ANC ≥100 cells/mcL, incidence of febrile neutropenia, incidence of documented infection, in-hospital all-cause mortality, and hospital length of stay.
We also compared the time to neutrophil engraftment in the SQ and IV groups based on transplant type (autologous vs allogeneic). Hospital length of stay was measured from the date of transplant to the date of discharge, and in-hospital all-cause mortality was defined as mortality that occurred during the hospital admission. All patients were hospitalized to receive their conditioning regimen and the stem-cell infusion.
G-CSF therapy was initiated between days 2 and 5 after stem-cell infusion at a dose of 5 mcg/kg once daily, rounded to the nearest vial size (300 mcg or 480 mcg), and therapy was continued until neutrophil engraftment. IV filgrastim was diluted in dextrose 5% to a concentration of 15 mcg/mL and given as a bolus injection through a central venous catheter over 15 to 30 minutes. Patients were assigned to each group according to the route of administration (SQ or IV) for >50% of administrations.
Statistical tests were performed using SPSS Statistics software (IBM; Armonk, NY). Continuous measurements were summarized using means and standard deviations. The chi square or Fisher’s exact test was used for nominal data and Student t-test for continuous data. A power analysis was not performed, because of the small patient population size. This study was approved by the hospital Institutional Review Board.
Results
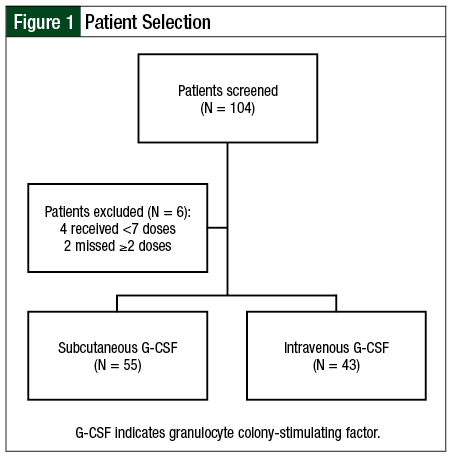
A total of 104 patients were screened for the study, and 98 patients met the inclusion criteria, grouped by the route of administration, which included 55 patients in the SQ group and 43 in the IV group (Figure 1). Overall, 35 patients had allogeneic HSCT and 63 had autologous HSCT. The baseline characteristics are summarized in Table 1.
G-CSF dosing was properly rounded in 89% of patients who received SQ administration and 95% of those who received IV. In some patients, the dosing was increased to twice daily based on physician judgment, if their condition did not show appropriate response, which usually consisted of a lack of neutrophil recovery at the average day of neutrophil engraftment at our institution. Of the study patients, 22% received twice daily dosing in the SQ arm and 26% in the IV arm.
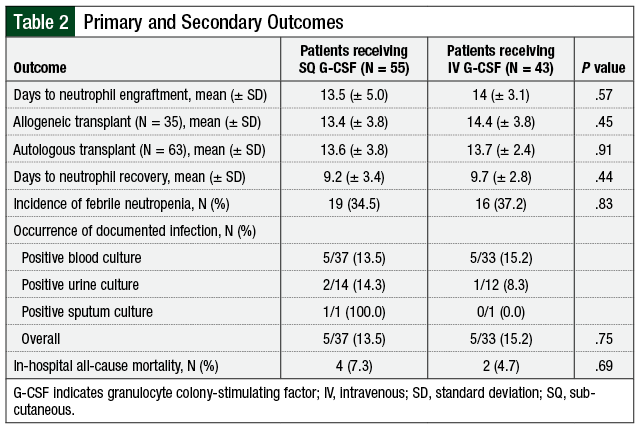
No difference was observed in time to neutrophil engraftment between the 2 groups, with a mean of 13.5 (± 5.0) days for SQ administration and 14 (± 3.1) days for IV administration (P = .57; Table 2). For patients who underwent allogeneic HSCT, the time to neutrophil engraftment was 1 day shorter among those who received SQ G-CSF, but this did not reach statistical significance (13.4 [± 3.8] days vs 14.4 [± 3.8] days; P = .45). For patients undergoing autologous transplant, no difference was observed in the time to neutrophil engraftment based on the route of administration (13.6 [± 3.8] days for SQ G-CSF and 13.7 [± 2.4] days for IV G-CSF; P = .91).
No difference in infection rates was observed between the 2 groups. Febrile neutropenia was reported in 34.5% of patients in the SQ group and 37.2% of patients in the IV group (P = .83). Of the patients who had cultures drawn while receiving G-CSF, 13.5% of patients receiving SQ therapy and 15.2% receiving IV therapy (P = .75) had positive cultures. The majority of positive cultures were blood cultures, which accounted for 5 of the 8 positive cultures in the SQ arm and 5 of the 6 positive cultures in the IV arm. Urine cultures were positive in 14.3% of patients in the SQ arm who had cultures collected compared with 8.3% of patients in the IV arm.
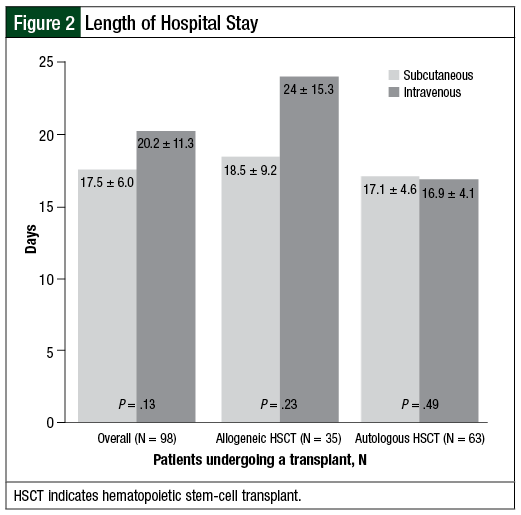
We did find differences in length of hospital stay between the 2 groups, but these differences were not statistically significant. Overall, the length of stay in patients undergoing allogeneic and autologous HSCT was 17.5 days in the SQ group and 20.2 days with IV administration (P = .13; Figure 2). This difference was most pronounced in the allogeneic transplant population, with a decreased length of hospital stay by 5.5 days in patients who received SQ G-CSF—18.5 days in the SQ arm and 24 days in the IV group (P = .23). No difference was seen between the 2 groups regarding in-hospital mortality, with rates of 7.2% and 4.7% in the SQ and IV groups, respectively.
Discussion
In this retrospective study comparing 2 routes of administration (SQ vs IV) of G-CSF in patients undergoing HSCT, we found no significant differences in the time to neutrophil engraftment, time to neutrophil recovery, febrile neutropenia rates, or length of hospital stay. We also found no significant differences after analyzing the study patients based on the transplant type.
To our knowledge, only 1 other study has assessed clinical outcomes related to the route of administration of G-CSF. Paul and colleagues compared SQ and IV administration in 120 hospitalized hematology oncology patients, of which 71 patients underwent either autologous or allogeneic transplant.19 The investigators found a longer time to neutropenia resolution in patients who received IV G-CSF compared with SQ G-CSF, and a mean 2.5-day advantage in time to neutropenia resolution when G-CSF was given as a SQ compared with IV administration.19 The subgroup of patients who underwent allogeneic HSCT had a significantly shorter time to stable neutrophil recovery associated with SQ G-CSF compared with IV, but no difference was found in autologous HSCT.19 There was no reported difference in the length of hospital stay; however, no subgroup analysis was conducted to evaluate the patients who underwent either autologous or allogeneic transplant.19
Our study showed no difference in time to neutrophil engraftment days overall, but patients who underwent allogeneic transplant were discharged 5.5 days sooner if they received SQ G-CSF compared with those who received IV. Although not statistically significant, this finding should be interpreted with caution, because of the greater number of patients undergoing allogeneic HSCT in the IV group compared with the SQ group. Larger trials are needed to determine if these differences truly exist in this patient population.
Limitations
This study must be viewed with its limitations. A main limitation is the relatively small sample size included in this study. There are also inherent limitations of conducting a retrospective study at a single center. Differences that may exist between the routes of administration of G-CSF may be difficult to elucidate in such a small population, especially when trying to discern variances in specific transplant type.
In addition, the patient population was heterogeneous; the disease type, conditioning regimens, donor source, and type of allogeneic transplant patients received may have affected the outcomes, independently of the route of administration of G-CSF and were not always balanced in the 2 arms of the study. Furthermore, the donor source and type of allogeneic transplant received were also not reported.
Finally, patients in the SQ and the IV arms received twice-daily dosing, which is greater than the recommended dose of G-CSF. Although balanced in the 2 arms, these doses are greater than the recommended weight-based dose of G-CSF.11
Conclusion
This is the first study to assess the impact of the route of administration of G-CSF in the patient population undergoing HSCT, which, despite the study’s small size, is an important contribution to the literature. Furthermore, few patients were excluded from the study, which may allow the results to be applied to a wider range of HSCT patients.
Prolonged neutropenia after transplant increases the risk for infectious complications and can contribute to morbidity and mortality. G-CSF is often used via IV or SQ administration to expedite the neutrophil engraftment process. Our findings suggest that the route of G-CSF administration after HSCT does not affect the outcomes, allowing for either route of administration to be used, based on provider and patient preferences. Larger confirmatory studies are needed to confirm our findings.
Author Disclosure Statement
Dr Ellingson, Dr Owolabi, Dr Wheelis, Dr Oliphant, and Dr Khaled have no conflicts of interest to report.
References
- Neupogen (filgrastim) injection, for subcutaneous or intravenous use [prescribing information]. Thousand Oaks, CA: Amgen; June 2018.
- Mossad SB, Longworth DL, Goormastic M, et al. Early infectious complications in autologous bone marrow transplantation: a review of 219 patients. Bone Marrow Transplant. 1996;18:265-271.
- Junghanss C, Marr KA. Infectious risks and outcomes after stem cell transplantation: are nonmyeloablative transplants changing the picture? Curr Opin Infect Dis. 2002;15:347-353.
- Klumpp TR, Mangan KF, Goldberg SL, et al. Granulocyte colony-stimulating factor accelerates neutrophil engraftment following peripheral-blood stem-cell transplantation: a prospective, randomized trial. J Clin Oncol.1995;13:1323-1327.
- McQuaker IG, Hunter AE, Pacey S, et al. Low-dose filgrastim significantly enhances neutrophil recovery following autologous peripheral-blood stem-cell transplantation in patients with lymphoproliferative disorders: evidence for clinical and economic benefit. J Clin Oncol.1997;15:451-457.
- Bishop MR, Tarantolo SR, Geller RB, et al. A randomized, double-blind trial of filgrastim (granulocyte colony-stimulating factor) versus placebo following allogeneic blood stem cell transplantation. Blood. 2000;96:80-85.
- Ozcan M, Ustün C, Akçaglayan E, et al. Recombinant human granulocyte colony-stimulating factor (rh-G-CSF) may accelerate hematopoietic recovery after HLA-identical sibling allogeneic peripheral blood stem cell transplantation. Bone Marrow Transplant. 2001;27:499-505.
- Khoury HJ, Loberiza FR Jr, Ringdén O, et al. Impact of posttransplantation G-CSF on outcomes of allogeneic hematopoietic stem cell transplantation. Blood. 2006;107:1712-1716.
- Trivedi M, Martinez S, Corringham S, et al. Optimal use of G-CSF administration after hematopoietic SCT. Bone Marrow Transplant. 2009;43:895-908.
- Smith TJ, Khatcheressian J, Lyman GH, et al. 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol.2006;24:3187-3205.
- Smith TJ, Bohlke K, Lyman GH, et al. Recommendations for the use of WBC growth factors: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol.2015;33:3199-3212.
- Trivedi M, Martinez S, Corringham S, et al. Review and revision of clinical practice of using G-CSF after autologous and allogeneic hematopoietic stem cell transplantation at UCSD. J Oncol Pharm Pract. 2011;17:85-90.
- Remberger M, Naseh N, Aschan J, et al. G-CSF given after haematopoietic stem cell transplantation using HLA-identical sibling donors is associated to a higher incidence of acute GVHD II-IV. Bone Marrow Transplant. 2003;32:217-223.
- Ringdén O, Hassan Z, Karlsson H, et al. Granulocyte colony-stimulating factor induced acute and chronic graft-versus-host disease. Transplantation. 2010;90:1022-1029.
- Ringdén O, Labopin M, Gorin NC, et al. Treatment with granulocyte colony-stimulating factor after allogeneic bone marrow transplantation for acute leukemia increases the risk of graft-versus-host disease and death: a study from the Acute Leukemia Working Party of the European Group for Blood and Marrow Transplantation. J Clin Oncol.2004;22:416-423.
- Dekker A, Bulley S, Beyene J, et al. Meta-analysis of randomized controlled trials of prophylactic granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor after autologous and allogeneic stem cell transplantation. J Clin Oncol.2006;24:5207-5215.
- Sugiura M, Yamamoto K, Sawada Y, Iga T. Pharmacokinetic/pharmacodynamic analysis of neutrophil proliferation induced by recombinant granulocyte colony-stimulating factor (rhG-CSF): comparison between intravenous and subcutaneous administration. Biol Pharm Bull. 1997;20:684-689.
- Wiczling P, Lowe P, Pigeolet E, et al. Population pharmacokinetic modelling of filgrastim in healthy adults following intravenous and subcutaneous administrations. Clin Pharmacokinet. 2009;48:817-826.
- Paul M, Ram R, Kugler E, et al. Subcutaneous versus intravenous granulocyte colony stimulating factor for the treatment of neutropenia in hospitalized hemato-oncological patients: randomized controlled trial. Am J Hematol. 2014;89:243-248.