Immunotherapy has transformed the treatment landscape for several cancers. Since 2014, there have been 7 immune checkpoint inhibitors for the PD-1/PD-L1 pathway approved by the FDA, including 4 for PD-1 (pembrolizumab, nivolumab, cemiplimab, and dostarlimab) and 3 for PD-L1 (atezolizumab, avelumab, and durvalumab).
Pembrolizumab is indicated for the treatment of a wide variety of oncologic diseases, including melanoma, non–small cell lung cancer (NSCLC), head and neck squamous cell cancer, classic Hodgkin lymphoma, primary mediastinal large B-cell lymphoma, microsatellite instability-high cancer, gastric cancer, esophageal cancer, urothelial carcinoma, cervical cancer, renal cell carcinoma, and others.1 Moreover, pembrolizumab is the first tissue- or site-agnostic agent indicated in patients with the microsatellite-high/DNA mismatch repair–deficient biomarker.2 According to pharmacokinetic modeling, pembrolizumab 400 mg every 6 weeks (Q6W) has a similar predicted exposure, including average concentration more than the dosing interval and steady-state concentration, to a dosage of 200 mg every 3 weeks (Q3W).3 In a study of patients with multiple cancer types who received pembrolizumab, the difference in predicted peak concentrations was 59% higher with 400-mg pembrolizumab dosed Q6W than with 200 mg dosed Q3W.3
In general, higher peak levels of medications may be associated with increased adverse events (AEs). Based on the findings of Lala and colleagues,3 on April 28, 2020, the FDA granted accelerated approval to pembrolizumab dosed at 400 mg Q6W, in addition to the previously approved dosage of 200 mg Q3W.4 However, the incidence of AEs with both regimens was not thoroughly described. Although the clinical safety data were consistent across the Q3W dosing regimens, Lala and colleagues only performed a predicted analysis of Q6W dosed pembrolizumab, which was expected to be similar to the Q3W dosing frequency.3
In this study, we conducted a retrospective chart review of patients who received pembrolizumab monotherapy to compare the incidence of AEs between the Q3W and Q6W dosing regimens.
Methods
This multicenter, retrospective chart review included patients aged ≥18 years who received at least one 200-mg or 400-mg dose of intravenous pembrolizumab monotherapy between April 2020 and October 2021. Patients were excluded from the study if they received systemic chemotherapy concurrently with pembrolizumab or if they switched dosing regimens more than 1 time to prevent overlapping AEs from 2 dosing regimens. The eligible patients were identified through electronic medical records (EMRs). The patients were assigned to 1 of 3 groups: pembrolizumab dosed Q3W, pembrolizumab dosed Q6W, or pembrolizumab switched from Q3W to Q6W dosing or from Q6W to Q3W dosing.
The study’s objective was to assess the rate of AEs related to pembrolizumab dosed at 400 mg Q6W versus at 200 mg Q3W. The primary end point for the study was the occurrence rate of treatment-related AEs with pembrolizumab monotherapy.
The secondary end points included the time to onset of treatment-related AEs, the reasons for treatment discontinuation, the incidence of all and severe treatment-related AEs, and the median duration of treatment. EMRs were used to access the providers’ notes, laboratory results, physical assessments, patients’ vitals, and the diagnostic evaluations. The AEs were categorized according to Common Terminology Criteria for Adverse Events Version 5.0.
Statistical Analysis
To discern the demographic and clinical characteristic differences between the groups, all categorical variables were summarized as frequencies and percentages and were compared between the arms by chi-square tests or Fisher’s exact test. Numeric variables were compared using t tests if they were normally distributed and reporting on the mean, and Kruskal–Wallis tests were used if they were not normally distributed and were reporting on the median. The categorical variables were compared using chi-square tests if comparing ≥2 groups, with Yate’s correction if there were zeroes in a cell, or Fisher’s exact test for comparing 2 variables with 2 factors and if the cell count was <5. The differences were considered significant if the P value was <.05. The statistics were computed in IBM SPSS Statistics version 28 (IBM Corporation).
After the completion of univariate comparisons, exploratory analyses were performed to predict if the patient was in the Q3W dose group or the Q6W dose group. Logistic regression was used when the outcome was 1 of the 2 study groups with Q3W dosing as the reference group. Predictors were selected based on whether there was a difference at the univariate level and by clinical relevance.
Results
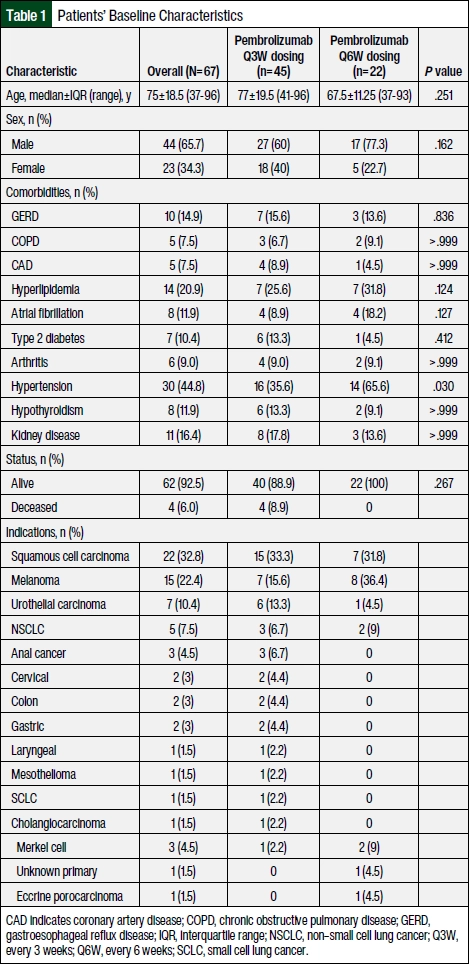
A total of 77 patients met the eligibility criteria, including 10 patients who switched dosing regimens. Of the total sample, 67 patients were considered for analyses to compare the Q3W dose group (N=45) with the Q6W dose group (N=22). Ten patients who switched dosing regimens were excluded from statistical analysis because of a small number of patients, and the results were reported separately. Table 1 summarizes the patients’ baseline characteristics. The median age of the overall study population was 75 years, the patients were predominantly men, and the 4 most common comorbidities were hypertension, hyperlipidemia, kidney disease, and gastroesophageal reflux disease. No significant differences were found between the cohorts in baseline characteristics other than in hypertension, in which there was a significantly higher percent of patients in the Q6W dose group who had hypertension than in the Q3W dose group (65.6% vs 35.6%, respectively; P=.03). The most common indications were squamous cell carcinoma, melanoma, urothelial carcinoma, and NSCLC.
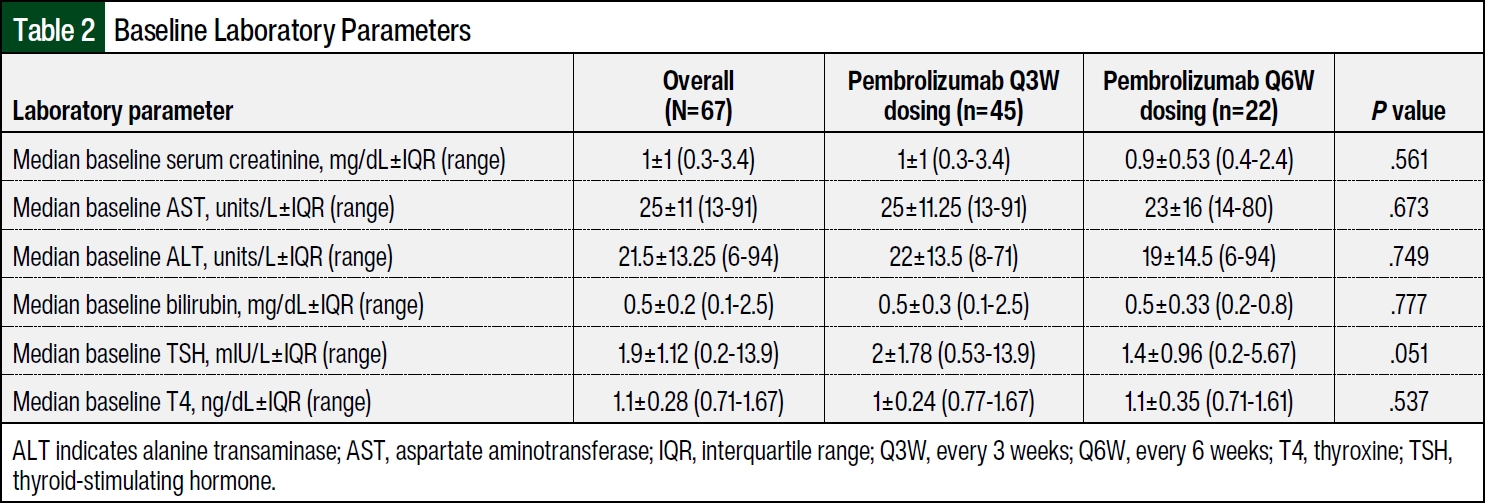
Table 2 summarizes the baseline laboratory parameters in the Q3W and Q6W dose groups. The baseline laboratory values were not significantly different between the groups. The median follow-up times were 12.5 months and 12 months in the Q3W and Q6W dose groups, respectively. Table 3 summarizes the previous chemotherapy treatments received and the reasons for treatment discontinuation. Although not statistically significant, fewer patients in the Q3W dose group discontinued pembrolizumab as a result of AEs than in the Q6W dose group (4.4% and 13.6%; P=.321). Significantly more patients in the Q3W dose group than in the Q6W dose group received previous chemotherapy (51.1% vs 18.2%, respectively; P=.018). Patients who received cisplatin before pembrolizumab were more likely to have Q3W dosing than Q6W dosing (27% vs 0%, respectively; P=.006). There were no significant differences between the 2 groups in indications or reasons for treatment discontinuation.
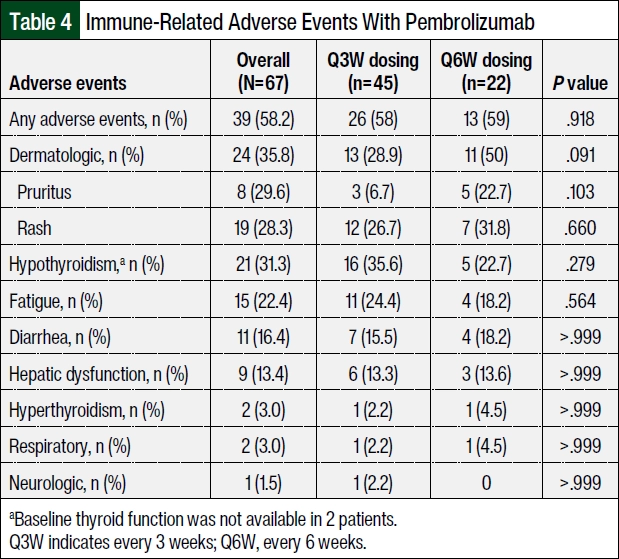
Overall, 39 (58.2%) of the 67 patients had any AE, of whom 26 (58%) and 13 (59%) of patients in the Q3W and Q6W dose groups, respectively, had an AE (Table 4). A total of 10 (14.9%) of the 67 study patients had grade 3 AEs. The grade 3 events included dermatologic events in 4.4% and 4.5% and diarrhea in 2.2% and 4.5% of patients in the Q3W and Q6W dose groups, respectively (Table 5). Table 4 and Table 5 compare the AEs between the 2 groups. No significant differences in the AEs between the groups were noted. Dermatologic events included rash and pruritus, and gastrointestinal events included diarrhea. Neurologic AEs included confusion, weakness, fatigue, and myalgias. Other AEs in the patients who received the Q3W dosing regimen included myalgia (n=2), nausea (n=2), arthralgia (n=1), back pain (n=1), dry mouth (n=1), esophagitis (n=1), and weakness (n=1). Other AEs in the patients who received the Q6W dosing regimen included headache (n=2), anhidrosis (n=1), and nausea (n=1). One patient in the Q6W dose group had a psoriasis flare at 11 weeks after starting treatment with pembrolizumab and had a newly diagnosed grade 2 arthralgia at 16 weeks.
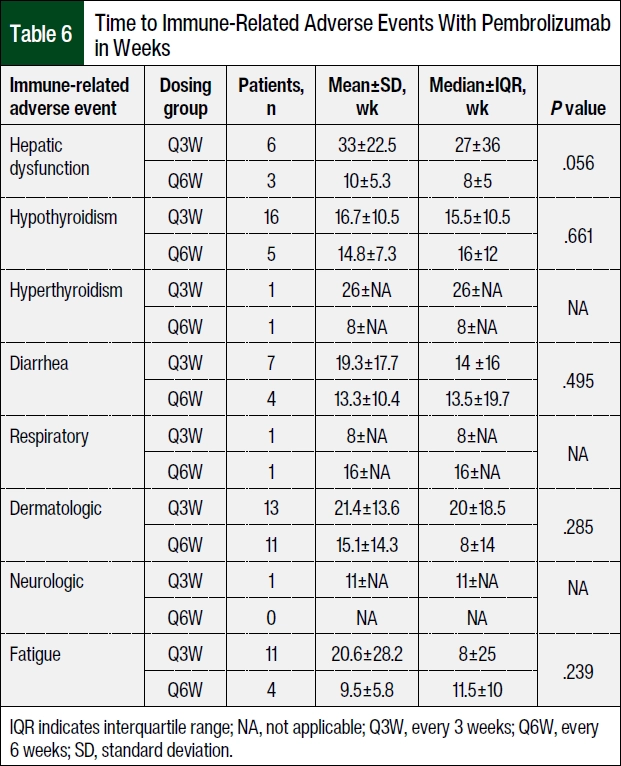
Table 6 summarizes the time to the AEs in weeks. The time to hepatic dysfunction immune-related AEs (irAEs) was approaching statistical significance, with the Q6W dose group having a shorter time to the onset of irAEs than the Q3W dose group (Δ=19; P=.056). The median times to hepatic dysfunction were 27±36 weeks and 8±5 weeks with the Q3W and Q6W dosing regimens, respectively. The mean time to hepatic dysfunction was 33±22.5 weeks and 10±5.3 weeks, respectively. Nonalcoholic fatty liver disease was observed before pembrolizumab treatment in 5 (7.4%) patients receiving the Q3W dosing regimen and in 7 (31.8%) patients receiving the Q6W dosing regimen (P=.472). Active alcohol use was reported in 14 (20.8%) and 6 (27.2%) patients in the Q3W and Q6W dose cohorts, respectively (P=.747).
Of the patients who had hepatic irAEs, 1 patient in the Q6W dose group had potential liver metastasis on imaging at baseline. All other differences in the times to events were not significantly different between the groups. The median times holding pembrolizumab were 8±24.5 weeks and 42±51.3 weeks in the Q3W and Q6W dose groups, respectively. The median number of pembrolizumab treatments was 14±11 (range, 1-57) weeks and 5±6 (range, 1-16) weeks in patients receiving the Q3W and Q6W dosing regimens, respectively. The difference in the median number of infusions can be accounted for by a longer treatment interval with the Q6W dosing regimen.
Treatment with pembrolizumab was discontinued as a result of AEs in 2 (4.4%) of the 45 patients in the Q3W dose group and in 1 (4.5%) of the 22 patients in the Q6W dose group. Systemic steroids were received for the treatment of immune-mediated reactions in 11.1% of patients in the Q3W dose group and in 4.5% of patients in the Q6W dose group. The median duration of systemic steroid use was 52.5 days (n=3 patients) and 48 days (n=5 patients) in the Q3W and Q6W dose cohorts, respectively. There were 2 patients in the Q3W dose group and 1 patient in the Q6W dose group for whom the time of steroid use could not be quantified. There was 1 patient who switched dosing regimens and received steroids for 175 days.
The exploratory analysis included baseline thyroid-stimulating hormone, cisplatin, chemotherapy before pembrolizumab, and time to hepatic dysfunction event. At the univariate level, previous chemotherapy significantly increased the odds of being included in the Q3W dose group by a factor of 4.3 compared with not receiving chemotherapy (95% confidence interval [CI], 1.35-16.73; P=.02). At the multivariate level, receiving previous chemotherapy significantly increased the odds of being included in the Q3W dose group by a factor of 7.72 when all other variables were held constant (95% CI, 1.77-44.89; P=.012).
A total of 10 patients switched dosage, with 9 patients switching from Q3W dosing to Q6W dosing and 1 patient switching from Q6W dosing to Q3W dosing. The median age was 76 years (range, 55-84), and 6 of the patients were women. The patients’ baseline aspartate aminotransferase, alanine aminotransferase, bilirubin, thyroid-stimulating hormone, and T4 were within normal limits. In patients who switched from Q3W dosing to Q6W dosing, the irAEs included dermatologic events (grade 1 in 4 patients and grade 2 in 1 patient), fatigue (grade 1 in 3 patients), hepatic events (grade 1 in 2 patients), and endocrine events (hypothyroidism in 1 patient and hyperthyroidism in 1 patient). In 1 patient who switched from Q6W to Q3W dosing, grade 1 fatigue was reported 2 weeks after the regimen switch. No irAEs led to treatment discontinuation.
Discussion
With the expansion of immunotherapy indications, it is estimated that up to 43.6% of patients with cancer in the United States are eligible to receive immune checkpoint inhibitors as of 2019.5 The side effects of treatment with immune checkpoint inhibitors may involve any organ or system, but the most prominent are dermatologic, gastrointestinal, hepatic, endocrine, or pulmonary AEs.6 Although the mechanism of the pathophysiology of irAEs is not fully elucidated, autoreactive T cells, autoantibodies, and inflammatory cytokines may play a role in this process.7
The dosing of pembrolizumab has evolved from weight-based (2 mg/kg Q3W) to flat dosing (200 mg Q3W and 400 mg Q6W) because it displays linear clearance at doses between 0.3 mg/kg and 10 mg/kg.8 The results of an exposure–safety analysis in patients with NSCLC in the phase 1 KEYNOTE-001 study showed that the incidence of AEs is similar across dosages of 2 mg/kg Q3W, 10 mg/kg Q3W, and 10 mg/kg Q2W.9 The results of our study are consistent with these findings, with the overall incidence of irAEs in our study being similar in the Q3W and Q6W dosing regimens.
The overall incidence of any-grade irAEs resulting from single-agent therapy with PD-1 or PD-L1 inhibitors was 26.82% in a meta-analysis in patients with various tumor types.10 In a meta-analysis of 17 phase 1 to 3 studies with pembrolizumab, the global incidence of irAEs was 18.5% (95% CI, 15.41-22.06).10 In a systematic review by Maughan and colleagues, the incidence rate of AEs with pembrolizumab in genitourinary cancers was 25%.11 Across immune checkpoint inhibitors, cutaneous AEs are the most common, occurring in up to 71.5% of patients, and comprise rash, pruritus, and vitiligo.6 Our study also demonstrates that the most frequent irAEs resulting from treatment with pembrolizumab are dermatologic events (ie, rash and pruritus) in 35.8% of patients. In the review by Maughan and colleagues, the incidence rates of serious (grade 3 or 4) irAEs were 16% to 22% in patients receiving single-agent immune checkpoint inhibitors.11 Our findings are similar to these results, with approximately 15% of patients having grade 3 AEs and no grade 4 or 5 AEs.
Since the start of this study, more recent literature was published comparing the incidences of irAEs with standard and extended interval dosing of pembrolizumab. The results of a retrospective review of the EMRs of 279 patients in Japan who received nivolumab or pembrolizumab for the treatment of advanced or metastatic solid tumors showed that 51.7% of the patients had irAEs.12 The results of our retrospective study demonstrated similar outcomes, with 58% and 59% of patients having irAEs in the Q3W and Q6W dose groups, respectively. Overall, the most common irAEs in the patients who received treatment in our study were dermatologic events (35.8%), hypothyroidism (31.3%), fatigue (22.4%), diarrhea (16.4%), and hepatic dysfunction (13.4%).
This is the first study to demonstrate a faster onset of hepatic irAEs with Q6W dosing. In a retrospective, observational study by Huffman and colleagues, patients with melanoma were diagnosed with hepatotoxicity after receiving ipilimumab, pembrolizumab, or the combination of ipilimumab and nivolumab.13 In Huffman and colleagues’ study, the median time to the onset of hepatic irAEs was 5 weeks to 12 weeks after the initiation of treatment.13 However, our results show a more prolonged median onset of hepatic irAEs at 8±5 weeks and 27±36 weeks with the Q6W and Q3W dosing regimens, respectively. Although not statistically significant, a higher proportion of our study’s patients in the Q6W dose group than in the Q3W dose group had nonalcoholic fatty liver disease at baseline or as reported on imaging, as well as active alcohol use, which could have been a confounding factor for a faster onset of hepatic irAEs with Q6W dosing. The shorter onset to hepatic irAEs in the study by Huffman and colleagues13 may be a result of irAEs being reported with PD-1 and PD-L1 as well as the CTLA-4 pathway, which are known to have a faster onset of irAEs in combination.14 Of note, previous studies have only included Q3W dosing of pembrolizumab11 or 2 mg/kg dosing every 3 weeks,13 which was used prior to the FDA approval of fixed dosing. To this date, we are not aware of any other published studies that have examined hepatic irAEs with Q6W dosing frequency since the FDA’s approval of pembrolizumab.
In a retrospective cohort study by Strohbehn and colleagues, only approximately 35% of patients adopted the Q6W dosing regimen for pembrolizumab, despite no difference in the time to treatment discontinuation between the Q3W and the Q6W dosing regimens.15 Similarly, in our study, more patients received the Q3W dosing regimen than the Q6W dosing regimen, even though both regimens were FDA approved at the time of the retrospective reviews. Previous chemotherapy significantly increased the patients’ odds of receiving pembrolizumab in a Q3W dosing regimen. Because many chemotherapy regimens have been delivered in a Q3W dosing regimen, it is possible that patients and providers were accustomed to this frequency.
One study that compared the molecular, preclinical, and early clinical characteristics of nivolumab and pembrolizumab reported that the receptor occupancy of nivolumab was as long as 57 days, which is similar to pembrolizumab.16 That study’s results led to an interest in extending the dosing frequency of pembrolizumab without increasing the dose.16 A recent study evaluated the extended duration of nivolumab treatment in patients with metastatic melanoma and Merkel cell carcinoma after 2 years of initial treatment.17 The progression- free survival rate was 100% in patients with a complete response, 89% in those with a partial response, and 50% in the patients with stable disease. Therefore, based on this research and the results of our study, the extended duration of pembrolizumab without increasing the dose could be evaluated.
Limitations
Because of this study’s retrospective design and lack of statistical power resulting from its inadequate patient sample size, it is challenging to distinguish irAEs between the groups. A short study period could explain the small sample sizes in each group. In addition, AEs could have been missed as a result of the chart review being limited to using the providers’ notes, imaging, and laboratory parameters from the EMR.
A total of 25 patients discontinued treatment with pembrolizumab, including 16 (35.6%) patients in the Q3W dose group and 9 (41%) patients in the Q6W dose group. The AEs were not being monitored after the discontinuation of treatment with pembrolizumab. Although performing such a review retrospectively is challenging, monitoring for AEs after the discontinuation of pembrolizumab treatment might have helped assess the late-onset irAEs. Because the incidence of nonalcoholic fatty liver disease was not factored in the statistical analysis, it could have contributed to a shorter time to onset of hepatic irAEs with the Q6W dosing regimen for pembrolizumab than the Q3W dosing regimen.
Last, although the exploratory analysis results were statistically significant, the wide CI for previous chemotherapy can be attributed to the amount of missing data and the small sample size.
Conclusion
Our study’s findings show that the incidence of any organ–related irAEs is similar between the Q3W and Q6W dosing regimens for pembrolizumab. However, although the incidence of hepatic irAEs was similar between the groups, the patients who received the Q6W dosing regimen had a faster time of onset to hepatic irAEs than those who received the Q3W dosing regimen. In addition, the patients who previously received chemotherapy were more likely to be assigned to the Q3W dosing regimen than the Q6W dosing regimen.
Funding Source
The authors would like to acknowledge the National Center for Advancing Translational Sciences of the National Institutes of Health Award number UL1TR002550.
Author Disclosure Statement
Dr Bollin is an advisor to Nektar Therapeutics. Dr Puglisi received research and grant support from Scripps Whittier Diabetes Institute and Scripps Health. Dr Kovalenko, Dr Garrett, and Dr Sikand have no conflicts of interest to report.
References
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck; March 2024. Accessed May 7, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/125514s160lbl.pdf
- Boyiadzis MM, Kirkwood JM, Marshall JL, et al. Significance and implications of FDA approval of pembrolizumab for biomarker-defined disease. J Immunother Cancer. 2018;6:35.
- Lala M, Li TR, de Alwis DP, et al. A six-weekly dosing schedule for pembrolizumab in patients with cancer based on evaluation using modelling and simulation. Eur J Cancer. 2020;131:68-75. Erratum in: Eur J Cancer. 2021;144:400.
- US Food and Drug Administration. FDA approves new dosing regimen for pembrolizumab. April 29, 2020. Accessed May 8, 2024. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-new-dosing-regimen-pembrolizumab
- Haslam A, Prasad V. Estimation of the percentage of US patients with cancer who are eligible for and respond to checkpoint inhibitor immunotherapy drugs. JAMA Netw Open. 2019;2:e192535.
- Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol. 2021;39:4073-4126. Erratum in: J Clin Oncol. 2022;40:315.
- Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158-168.
- Centanni M, Moes DJAR, Trocóniz IF, et al. Clinical pharmacokinetics and pharmacodynamics of immune checkpoint inhibitors. Clin Pharmacokinet. 2019;58:835-857.
- Chatterjee M, Turner DC, Felip E, et al. Systematic evaluation of pembrolizumab dosing in patients with advanced non-small-cell lung cancer. Ann Oncol. 2016;27:1291-1298.
- Wang PF, Chen Y, Song SY, et al. Immune-related adverse events associated with anti-PD-1/PD-L1 treatment for malignancies: a meta-analysis. Front Pharmacol. 2017;8:730.
- Maughan BL, Bailey E, Gill DM, Agarwal N. Incidence of immune-related adverse events with program death receptor-1- and program death receptor-1 ligand-directed therapies in genitourinary cancers. Front Oncol. 2017;7:56.
- Morimoto K, Yamada T, Morimoto Y, et al. A real-world study on the safety of the extended dosing schedule for nivolumab and pembrolizumab in patients with solid tumors. Int Immunopharmacol. 2022;108:108775. Erratum in: Int Immunopharmacol. 2022;112:109232.
- Huffman BM, Kottschade LA, Kamath PS, Markovic SN. Hepatotoxicity after immune checkpoint inhibitor therapy in melanoma: natural progression and management. Am J Clin Oncol. 2018;41:760-765.
- De Martin E, Michot JM, Papouin B, et al. Characterization of liver injury induced by cancer immunotherapy using immune checkpoint inhibitors. J Hepatol. 2018;68:1181-1190.
- Strohbehn GW, Holleman R, Burns J, et al. Adoption of extended-interval dosing of single-agent pembrolizumab and comparative effectiveness vs standard dosing in time-to-treatment discontinuation. JAMA Oncol. 2022;8:1663-1667.
- Fessas P, Lee H, Ikemizu S, Janowitz T. A molecular and preclinical comparison of the PD-1-targeted T-cell checkpoint inhibitors nivolumab and pembrolizumab. Semin Oncol. 2017;44:136-140.
- Tachiki LML, Williams Silva K, Hippe DS, et al. Extended duration of anti-PD-1 therapy, using reduced frequency dosing, in patients with advanced melanoma and Merkel cell carcinoma. J Clin Oncol. 2022;40(suppl 16):2588.