Connecting Science to Practice
Cisplatin-based chemotherapy, the cornerstone of management
of a wide range of cancers, can elicit potent cytotoxic
and radiosensitizing effects, but its efficacy is limited by adverse
events (AEs). Mannitol had been used as a part of the
cisplatin regimen to reduce cisplatin-induced nephrotoxicity
(CIN), but this combination has fallen out of favor as a
result of a brief shortage of mannitol and conflicting data.
To explore this issue, we evaluated the role of mannitol on
the reduction of CIN and on the cumulative cisplatin dose
(CCD); we also assessed practice pattern changes after the
mannitol shortage of 2011. We found that the use of mannitol
was associated with less CIN and a higher CCD. These
data suggest that there is a need to incorporate mannitol
into cisplatin-based regimens. Our survey of hospital institutions
suggests that the use of mannitol to reduce CIN has
largely fallen out of practice. Our data are compelling and
highlight the need for additional prospective studies to
evaluate the risk for nephrotoxicity with cisplatin plus mannitol
therapy versus with cisplatin alone. Mannitol may
have a significant role in reducing the risk for CIN and deserves
further investigation.
Cisplatin-based chemotherapy remains the cornerstone of management of a wide range of cancers.1-4 Cisplatin elicits potent cytotoxic and radiosensitizing effects by cross-linking DNA in rapidly dividing cells, which results in cell death1; however, cisplatin’s dose-dependent efficacy is limited by many adverse events (AEs).5 Cisplatin-induced nausea can be well managed through the use of highly effective antiemetic drugs; however, other AEs, such as electrolyte disturbances, neurotoxicity, and ototoxicity, are frequently irreversible.6-10 Nephrotoxicity, which is frequently defined as a ≥25% decline in estimated glomerular filtration rate (eGFR) from baseline,11 is frequently encountered with cisplatin treatment and is now the most common dose-limiting AE of cisplatin.1,12,13
Cisplatin-induced nephrotoxicity (CIN) is dose- and schedule-dependent, and is associated with acute and chronic kidney damage and a significant risk for progression to chronic kidney disease and increased mortality.6,14,15 Up to 30% of patients who receive cisplatin therapy have an acute kidney injury (AKI), typically nonoliguric, which can be seen even after a single dose.6 The incidence of CIN may be as high as 40% to 50% in patients receiving daily cisplatin regimens administered over consecutive days.16
Cisplatin is renally excreted, and CIN occurs as a result of the accumulation of cisplatin in the proximal renal tubules, increasing the renal concentration up to 5 times as high as the serum concentration, in conjunction with the baseline susceptibility of mitochondrial DNA to cisplatin damage.2,11,17 Mitochondrial DNA has less efficient repair mechanisms, making it more susceptible to cisplatin-induced damage than nuclear DNA.6 Consequently, the disruption of mitochondrial energetics leads to direct necrosis of the renal tubular cells.18
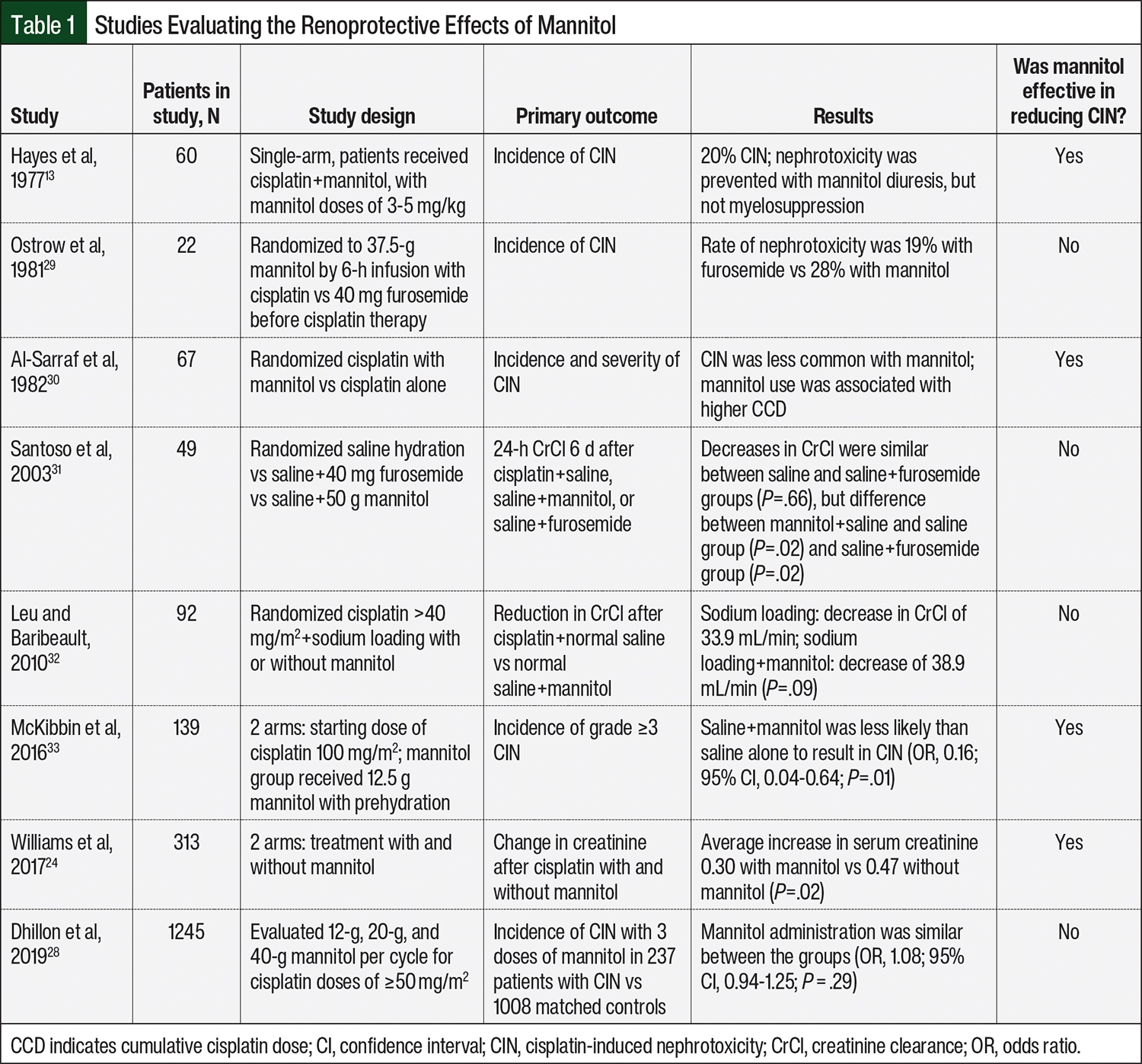
Earlier studies have investigated the risk factors for and preventive measures to reduce CIN.19,20 Intravenous (IV) hydration, when used in conjunction with cisplatin, reduces the half-life of cisplatin as well as the proximal tubule transit time and is the primary intervention to prevent CIN.13,21,22 Increasing the osmolarity of the glomerular filtrate, promoting the excretion of water, and decreasing the reabsorption of solutes also reduces CIN.23 Mannitol, a sugar alcohol, is an osmotic diuretic that, in conjunction with hydration, reduces renal cisplatin concentrations and has traditionally been used to prevent nephrotoxicity.24,25 Mannitol by itself is considered nephrotoxic because of its effect on the proximal tubule, causing the vacuolization of tubular cells, vasoconstriction of the afferent arteriole, and intense diuresis after treatment with mannitol.26,27 It is unclear if treatment with mannitol truly affects the incidence of CIN, because studies that have evaluated the role of mannitol in mitigating CIN have been low-powered with inconsistent mannitol dosing and, therefore, have yielded mixed results (Table 1).13,24,25,28-33
A national shortage of mannitol in 2011 resulted in the limited availability of mannitol for routine use. This limited accessibility, as well as inconsistent data supporting its use, were potentially the impetus for a shift in practice habits away from using mannitol as a presumed renoprotective agent. We sought to examine this temporary drug shortage to evaluate the effect of mannitol on CIN. We hypothesized that the administration of mannitol would be associated with a lower incidence of CIN. In addition, we surveyed several institutions to determine their evolving practice patterns regarding treatment with mannitol before and after the initial shortage of the drug.
Methods
This retrospective-cohort study examined all patients who received cisplatin therapy from 2006 to 2012 at Froedtert & The Medical College of Wisconsin. Institutional review board approval was obtained and followed the Declaration of Helsinki principles. All patients who received cisplatin were identified by a search by our electronic chemotherapy management software, EPIC Beacon (EPIC Systems; Verona, WI). Patients who were diagnosed with multiple myeloma were excluded as a result of potential renal dysfunction secondary to their multiple myeloma diagnosis. The patients’ baseline characteristics, oncologic diagnosis, cisplatin dosing, and mannitol administration at each treatment cycle were collected, and their eGFR was calculated using the CKD-EPI Creatinine Equation. Per institutional standard practice, cisplatin was diluted in 500 or 1000 mL of sodium chloride 0.9%, with a maximum cisplatin concentration of 0.4 mg/mL, and was administered over 1 hour. When mannitol was administered, it was added to the cisplatin. The patients received IV hydration with at least 1L of normal saline before the administration of cisplatin.
The study’s primary objective was to determine if adding mannitol to cisplatin regimens resulted in a decrease in nephrotoxicity. Nephrotoxicity was defined as an occurrence of AKI. AKI was defined as a ≥25% decrease in eGFR per the RIFLE criteria that occurred at any point during cisplatin therapy.34 Our secondary objective was to determine if patients who received cisplatin plus mannitol were able to receive a higher cumulative cisplatin dose (CCD) as defined by the cumulative dose of cisplatin received by each patient over all treatment cycles.
Statistical Analysis
Statistical analysis was performed using SAS 9.4 (SAS Institute; Cary, NC). The patients were categorized into 1 of 2 groups—cisplatin plus mannitol or cisplatin alone—based on the use of mannitol in the first cycle. Univariate between-group comparisons were performed using the chi-square test for categorical variables and the Wilcoxon rank-sum test for continuous outcomes, including cisplatin dose. Survival analysis techniques were used to evaluate the relationship between CCD and nephrotoxicity and its modification by mannitol administration.
The first occurrence of nephrotoxicity was the event variable in this analysis, whereas the cumulative dose received by the patient before nephrotoxicity was considered the time variable. The patients without nephrotoxicity were censored at the total cumulative dose received. The unadjusted cumulative incidence of nephrotoxicity for a given CCD was computed for hypothetical patients receiving mannitol at every cycle or in none of the cycles using Cox regression that was stratified by mannitol use status. Cox proportional hazards regression was performed to obtain the estimates of the effect of mannitol, and were adjusted for sex, race, age, diagnostic group, and baseline eGFR. Robust sandwich variance estimates were used to incorporate dependence because of treatment occasions with and without mannitol from the same patient.
Sensitivity Analysis
The primary analysis defined AKI as a 25% reduction in eGFR from baseline. The Cox regression analyses were repeated, with cutoffs ranging from a 10% decrease to a 60% decrease to evaluate the sensitivity of the conclusions.
The survey was sent electronically to a hospital pharmacy listserv. These hospital pharmacies were nationwide and included academic and community-based institutions with cancer centers. A total of 41 institutions completed a 4-question survey regarding mannitol use in cisplatin therapy. The questions included:
- Does your organization currently use mannitol in combination with cisplatin for renal protection?
- Did your organization use mannitol in combination with cisplatin for renal protection before the mannitol drug shortage (circa 2011)?
- Did your organization stop using mannitol as a result of the drug shortage?
- Postshortage, did your organization add mannitol back into cisplatin infusions?
The survey responses were compiled using an Excel spreadsheet. The responses were then compared using percentages without formal statistical analysis. Our primary goal was to determine if there was a change in the use of mannitol administration for renal protection with cisplatin chemotherapy after the brief shortage in 2011.
Results
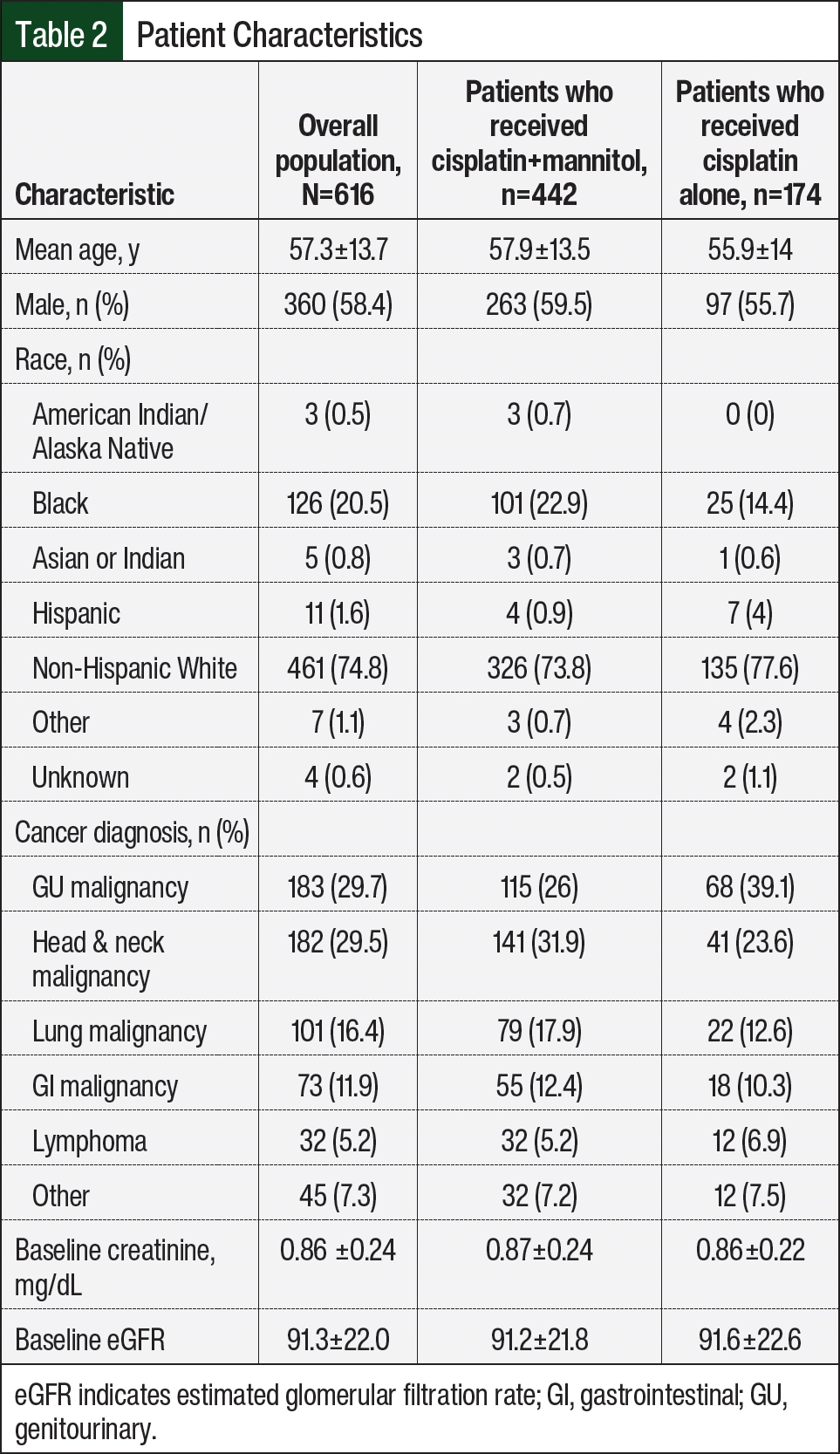
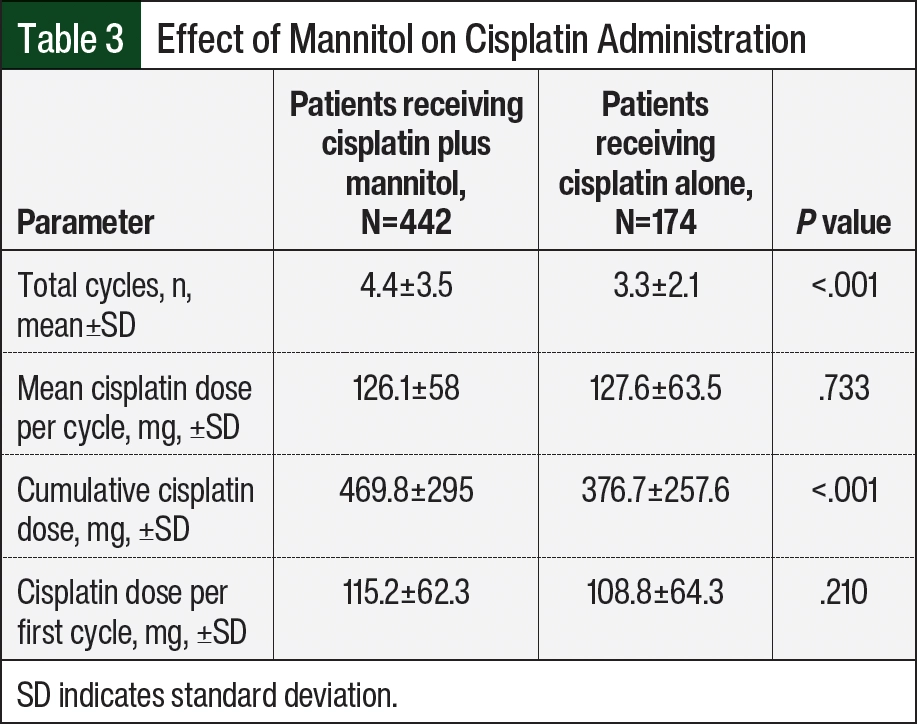
The characteristics of the patient cohorts are listed in Table 2. A total of 616 patients with a mean age of 57.3±13.7 years were identified. The patients in this study had a wide variety of oncologic diagnoses; genitourinary and head and neck malignancies were the most common oncologic diagnosis, with each representing approximately 30% of the cohort. In all, 442 (approximately 72%) of the patients received concurrent treatment with cisplatin and mannitol in the first cycle, whereas 174 (approximately 28%) received cisplatin without mannitol. Among the patients who received concurrent cisplatin and mannitol in the first cycle, 371 (approximately 84%) received mannitol in all cycles; among the patients who did not receive concurrent cisplatin and mannitol in the first cycle, 153 (88%) never received concurrent cisplatin and mannitol. Treatment with mannitol was discontinued abruptly in November 2011 secondary to this shortage, although treatment tended to be more uniform before or after the shortage. Although the distributions by race and diagnosis were unbalanced between the groups, the patients were similar in terms of oncologic diagnosis, baseline renal function, and intended cisplatin dosing, as is evidenced by the dose in the first cycle (Table 2 and Table 3).
The patients who received cisplatin plus mannitol received more cycles of cisplatin therapy (4.4±3.5 vs 3.3±2.1 cycles, respectively; P<.001) and had a higher mean CCD than those who received cisplatin alone (469.8±295 mg versus 376.7±258 mg, respectively; P<.001; Table 3). The patients who received cisplatin plus mannitol had a lower incidence of nephrotoxicity than the patients who received cisplatin alone (26.5% vs 35.1%, respectively; P=.034).
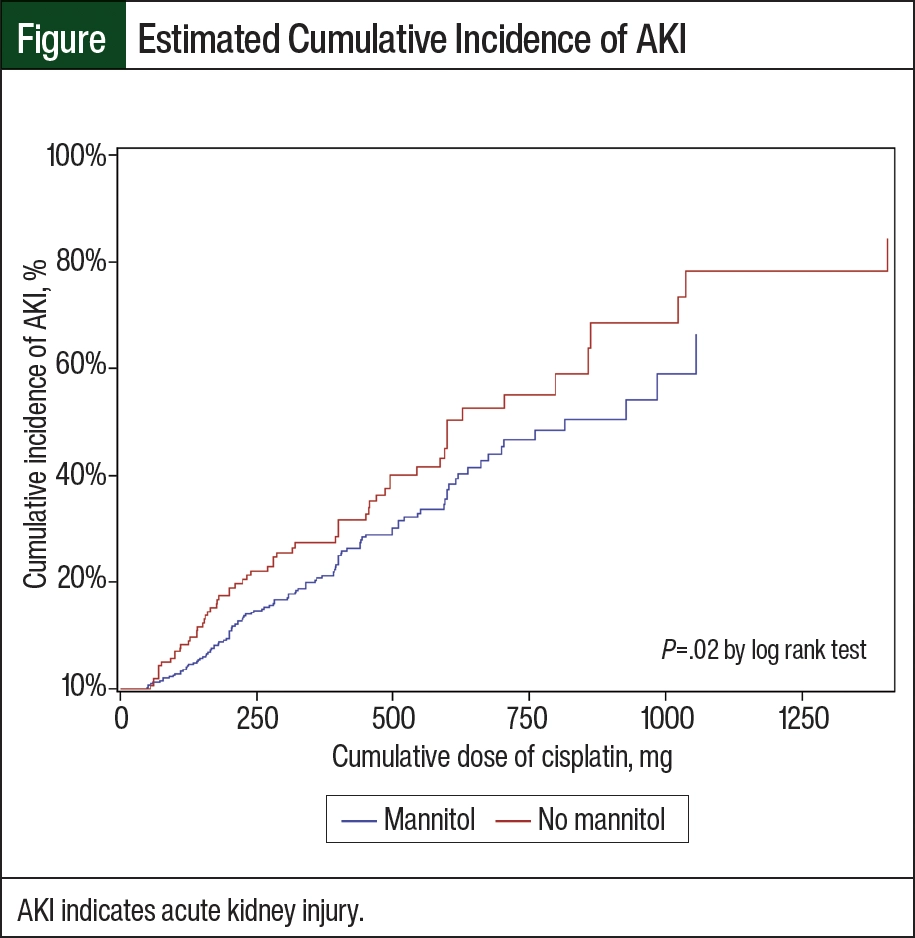
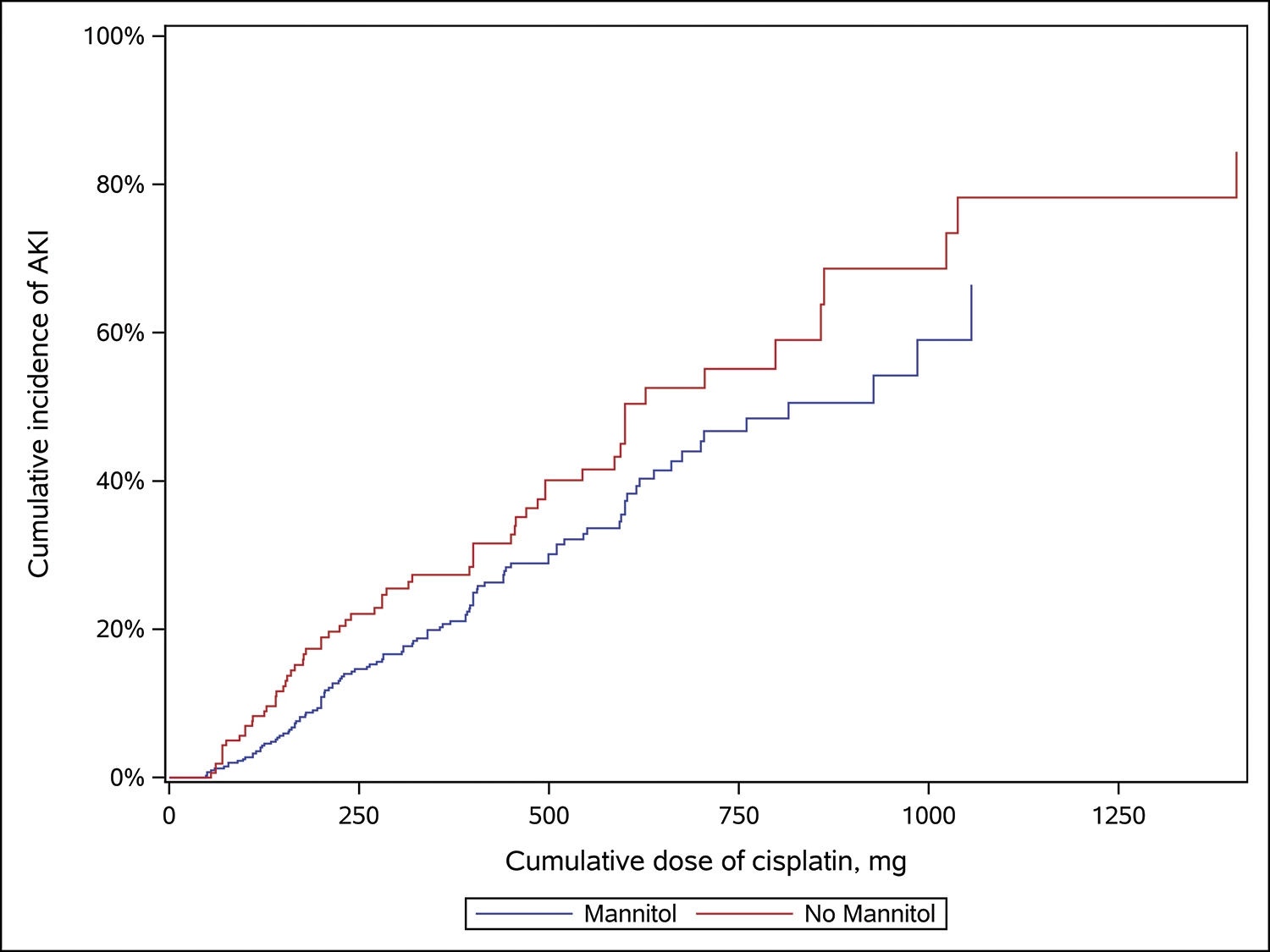
The estimated cumulative incidence of AKI as a function of current CCD for patients receiving mannitol every cycle versus in none of the cycles is shown in the Figure. For a 500-mg CCD, 31.5% of patients receiving cisplatin plus mannitol had AKI (95% confidence interval [CI], 25.5-37.0), whereas 41.6% of those receiving only cisplatin had AKI (95% CI, 31.3-50.3). A univariate analysis demonstrated that treatment with cisplatin plus mannitol was associated with a 30% reduction in the risk for CIN versus cisplatin alone (hazard ratio [HR], 0.70; 95% CI, 0.51-0.94; P=.02). A multivariable analysis adjusting for baseline eGFR, age, sex, race, and oncologic diagnosis demonstrated that treatment with mannitol was associated with a 37% percent reduction in the risk of CIN (HR, 0.63; 95% CI, 0.46-0.87; P=.004) compared with patients receiving cisplatin alone.
Hispanic ethnicity was associated with an increased risk for CIN compared with non-Hispanic White ethnicity (HR, 2.5; 95% CI, 1.11-5.83; P=.03; Table 4). Older age (HR, 1.2 per decade; 95% CI, 1.06-1.37; P=.005) and female sex (HR, 1.80; 95% CI, 1.31-2.46; P=.0003) were also identified as significant risk factors for CIN.
In all, 41 institutions responded to the survey. A total of 31 (76%) institutions were using mannitol to reduce the risk for CIN before the shortage in 2011, with 10 (24%) institutions not previously using mannitol. Of the 31 institutions that had used mannitol before the shortage in 2011, 15 (approximately 48%) had stopped using it by the time of the survey. Of those institutions that stopped using mannitol, 11 (approximately 73%) stated that they did so as a result of the drug shortage. Only 5 (approximately 12%) institutions that were surveyed had incorporated mannitol back into cisplatin infusions after the shortage as of the time of the survey.
Discussion
Decades after its introduction into clinical medicine, cisplatin remains a crucial component of treatment for a wide variety of cancers, including head and neck malignancies and genitourinary cancers. Nephrotoxicity remains a dose-limiting AE. Unfortunately, approaches to ameliorate the rate of CIN, including treatment with mannitol, have yielded conflicting results. Our study demonstrates that in patients with cancer who received cisplatin, treatment with mannitol was associated with a significant reduction in the risk for nephrotoxicity. The decreased risk for nephrotoxicity was independently associated with mannitol treatment when adjusting for baseline eGFR, age, sex, race, and disease type.
Our findings contrast those of the 2019 study by Dhillon and colleagues that did not find a significant difference in the incidence of CIN between cisplatin plus mannitol and cisplatin alone.28 However, the univariable analysis by Dhillon and colleagues in 2019 analyzed the incidence of nephrotoxicity using 3 different doses of mannitol rather than the overall effectiveness when contrasted as a group. The subgroup analysis in this study showed benefit of mannitol administration when received at doses of 50 mg/m2 per cycle.28
Our data demonstrate that the probability of AKI occurred at a higher CCD in those patients who received mannitol versus patients who received cisplatin alone. The patients who received cisplatin plus mannitol received more cycles of therapy (thus, more administrations of cisplatin) and had a higher mean CCD than those patients who did not receive mannitol therapy. This is critical because previous studies have demonstrated improved survival outcomes with a higher CCD.35-37 The study by Gundog and colleagues also demonstrated that the number of cycles of cisplatin delivered was an independent prognostic factor of outcome.38
The incidence of AKI existed for any eGFR percentage decrease of more than 25% when sensitivity analysis was performed (data not shown). We selected a 25% decrease as the cutoff for nephrotoxicity because this was in line with clinical practice guidelines.38 Still, this difference existed for any eGFR percentage decrease greater than 25%, suggesting an actual renoprotective effect of mannitol.
Our survey of hospital institutions suggests that the use of mannitol to reduce CIN has largely fallen out of practice. At the time of our survey, only 37% of the surveyed institutions actively prescribed mannitol in conjunction with cisplatin therapy. Only approximately 12% of the institutions that we surveyed had reincorporated mannitol into treatment after the shortage. Our data are compelling and highlight the need for additional prospective studies to evaluate the risk for nephrotoxicity with cisplatin plus mannitol therapy versus with cisplatin alone. Mannitol may have a significant role in reducing the risk for CIN and deserves further investigation.
Limitations
This study includes a large cohort of more than 600 patients with various oncologic diagnoses. Our study does have several limitations, such as its retrospective design, only including data sourced from a single institution, the heterogeneity of the patient population and cisplatin regimens, a lack of randomization, and the possible confounding from other etiologies of AKI. Also, the data were not collected on the concurrent administration of medications that can result in AKI (eg, nonsteroidal anti-inflammatory drugs) or augment diuresis (eg, hydrochlorothiazide and loop diuretics). Despite these limitations, this study shows that mannitol may reduce the risk for nephrotoxicity and supports the need for further investigation into the inclusion of mannitol in cisplatin regimens.
Conclusion
The inclusion of mannitol in treatment with cisplatin was associated with a lower incidence of AKI, a higher number of cycles of cisplatin therapy received, and a higher mean CCD administered than cisplatin therapy alone. Our survey of institutions shows that the use of mannitol in cisplatin-containing regimens has fallen out of practice. Still, our data indicate that treatment with mannitol may be renoprotective, allowing for a higher CCD and, thereby, better outcomes. The reintroduction of mannitol into cisplatin-based chemotherapeutic regimens should be further investigated.
Acknowledgments
The authors would like to acknowledge the work of Julie Karpinski, PharmD, Sridevi Ramalingam, MD, and Chitra S. Bhosekar, MD, in the preparation of this manuscript.
Author Disclosure Statement
Dr Kilari is on the speakers bureau for Aveo Oncology, Exelixis, Janssen, and Seagen and is a consultant to Aveo Oncology, Exelixis, MJH Life Sciences, Myovant, and Pfizer; Dr Wong has received research funding from Hookipa Pharma and Merck; and Dr Berlin, Dr Szabo, Dr Bhasin-Chhabra, Dr Nelson, Dr Giever, and Dr Pawlenty have no conflicts of interest to report.
References
- Hanigan MH, Devarajan P. Cisplatin nephrotoxicity: molecular mechanisms. Cancer Ther. 2003;1:47-61.
- Morris M, Eifel PJ, Lu J, et al. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. N Engl J Med. 1999;340:1137-1143.
- Arriagada R, Bergman B, Dunant A, et al. Cisplatin-based adjuvant chemotherapy in patients with completely resected non-small-cell lung cancer. N Engl J Med. 2004;350:351-360.
- Zhang Y, Chen L, Hu GQ, et al. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N Engl J Med. 2019;381:1124-1135.
- Percie du Sert N, Rudd JA, Apfel CC, Andrews PLR. Cisplatin-induced emesis: systematic review and meta-analysis of the ferret model and the effects of 5-HT3 receptor antagonists. Cancer Chemother Pharmacol. 2011;67:667-686.
- Miller RP, Tadagavadi RK, Ramesh G, Reeves WB. Mechanisms of cisplatin nephrotoxicity. Toxins (Basel). 2010;2:2490-2518.
- Madias NE, Harrington JT. Platinum nephrotoxicity. Am J Med. 1978;65:307-314.
- Kanat O, Ertas H, Caner B. Platinum-induced neurotoxicity: a review of possible mechanisms. World J Clin Oncol. 2017;8:329-335.
- Sheth S, Mukherjea D, Rybak LP, Ramkumar V. Mechanisms of cisplatin-induced ototoxicity and otoprotection. Front Cell Neurosci. 2017;11:338.
- Astolfi L, Ghiselli S, Guaran V, et al. Correlation of adverse effects of cisplatin administration in patients affected by solid tumours: a retrospective evaluation. Oncol Rep. 2013;29:1285-1292.
- Coresh J, Turin TC, Matsushita K, et al. Decline in estimated glomerular filtration rate and subsequent risk of end-stage renal disease and mortality. JAMA. 2014;311:2518-2531.
- Wei Q, Dong G, Franklin J, Dong Z. The pathological role of Bax in cisplatin nephrotoxicity. Kidney Int. 2007;72:53-62.
- Hayes DM, Cvitkovic E, Golbey RB, et al. High dose cis-platinum diammine dichloride: amelioration of renal toxicity by mannitol diuresis. Cancer. 1977;39:1372-1381.
- Silver SA, Harel Z, McArthur E, et al. Causes of death after a hospitalization with AKI. J Am Soc Nephrol. 2018;29:1001-1010.
- Holditch SJ, Brown CN, Lombardi AM, et al. Recent advances in models, mechanisms, biomarkers, and interventions in cisplatin-induced acute kidney injury. Int J Mol Sci. 2019;20:3011.
- Imamura Y, Kiyota N, Tahara M, et al. Risk prediction model for cisplatin-induced acute kidney injury in patients with head and neck cancer receiving chemoradiotherapy: a re-analysis of a phase II/III JCOG1008 trial. Oral Oncol. 2024;154:106868.
- Yao X, Panichpisal K, Kurtzman N, Nugent K. Cisplatin nephrotoxicity: a review. Am J Med Sci. 2007;334:115-124.
- Linkermann A, Chen G, Dong G, et al. Regulated cell death in AKI. J Am Soc Nephrol. 2014;25:2689-2701.
- Almanric K, Marceau N, Cantin A, Bertin É. Risk factors for nephrotoxicity associated with cisplatin. Can J Hosp Pharm. 2017;70:99-106.
- Hayati F, Hossainzadeh M, Shayanpour S, et al. Prevention of cisplatin nephrotoxicity. J Nephropharmacol. 2016;5:57-60.
- Frick GA, Ballentine R, Driever CW, Kramer WG. Renal excretion kinetics of high-dose cis-dichlorodiammineplatinum(II) administered with hydration and mannitol diuresis. Cancer Treat Rep. 1979;63:13-16.
- Daugaard G, Abildgaard U. Cisplatin nephrotoxicity. A review. Cancer Chemother Pharmacol. 1989;25:1-9.
- Morgan KP, Buie LW, Savage SW. The role of mannitol as a nephroprotectant in patients receiving cisplatin therapy. Ann Pharmacother. 2012;46:276-281.
- Williams RP Jr, Ferlas BW, Morales PC, Kurtzweil AJ. Mannitol for the prevention of cisplatin-induced nephrotoxicity: a retrospective comparison of hydration plus mannitol versus hydration alone in inpatient and outpatient regimens at a large academic medical center. J Oncol Pharm Pract. 2017;23:422-428.
- Morgan KP, Snavely AC, Wind LS, et al. Rates of renal toxicity in cancer patients receiving cisplatin with and without mannitol. Ann Pharmacother. 2014;48:863-869.
- Rachman A, Wafa S, Nugroho P, Koesnoe S. The effect of mannitol addition to hydration on acute kidney injury event after high dose cisplatin chemotherapy: an ambispective cohort study. BMC Cancer. 2022;22:395.
- Lin SY, Tang SC, Tsai LK, et al. Incidence and risk factors for acute kidney injury following mannitol infusion in patients with acute stroke: a retrospective cohort study. Medicine (Baltimore). 2015;94:e2032.
- Dhillon P, Amir E, Lo M, et al. A case-control study analyzing mannitol dosing for prevention of cisplatin-induced acute nephrotoxicity. J Oncol Pharm Pract. 2019;25:875-883.
- Ostrow S, Egorin MJ, Hahn D, et al. High-dose cisplatin therapy using mannitol versus furosemide diuresis: comparative pharmacokinetics and toxicity. Cancer Treat Rep. 1981;65:73-78.
- Al-Sarraf M, Fletcher W, Oishi N, et al. Cisplatin hydration with and without mannitol diuresis in refractory disseminated malignant melanoma: a southwest oncology group study. Cancer Treat Rep. 1982;66:31-35.
- Santoso JT, Lucci JA 3rd, Coleman RL, et al. Saline, mannitol, and furosemide hydration in acute cisplatin nephrotoxicity: a randomized trial. Cancer Chemother Pharmacol. 2003;52:13-18.
- Leu L, Baribeault D. A comparison of the rates of cisplatin (cDDP)-induced nephrotoxicity associated with sodium loading or sodium loading with forced diuresis as a preventative measure. J Oncol Pharm Pract. 2010;16:167-171.
- McKibbin T, Cheng LL, Kim S, et al. Mannitol to prevent cisplatin-induced nephrotoxicity in patients with squamous cell cancer of the head and neck (SCCHN) receiving concurrent therapy. Support Care Cancer. 2016;24:1789-1793.
- Lopes JA, Jorge S. The RIFLE and AKIN classifications for acute kidney injury: a critical and comprehensive review. Clin Kidney J. 2013;6:8-14.
- Hesketh PJ, Tansan S, Caguioa PB, et al. Treatment of advanced non-small cell lung cancer with very high-dose cisplatin combined with etoposide and mitomycin C. Cancer. 1993;71:717-720.
- Gundog M, Basaran H, Bozkurt O, Eroglu C. A comparison of cisplatin cumulative dose and cisplatin schedule in patients treated with concurrent chemo-radiotherapy in nasopharyngeal carcinoma. Braz J Otorhinolaryngol. 2020;86:676-686.
- Tjan-Heijnen VCG, Wagener DJT, Postmus PE. An analysis of chemotherapy dose and dose-intensity in small-cell lung cancer: lessons to be drawn. Ann Oncol. 2002;13:1519-1530.
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1-150.