Connecting Science to Practice
Prolonged thrombocytopenia after hematopoietic stem-cell
transplantation (HSCT) is a risk factor for increased morbidity
and mortality. Eltrombopag has been proposed as a
treatment option in patients with post-HSCT thrombocytopenia,
but current evidence is limited to studies with small
sample sizes and variable dosing, duration, and response
rates. This retrospective review included 67 patients who
received eltrombopag for thrombocytopenia after allogeneic
stem-cell transplant. Overall, 58% of patients achieved a
platelet response (defined as platelet level ≥50,000/μL), with
a median time to response of 28 days. Patients with post-
HSCT thrombocytopenia may benefit from treatment with
eltrombopag, but platelet response may take weeks to reach.
Hematopoietic stem-cell transplantation (HSCT) is widely used as a treatment for nonmalignant and malignant hematologic disorders. One risk factor for increased morbidity and mortality after HSCT is prolonged thrombocytopenia, which occurs in up to 37% of patients who receive HSCT.1,2 The etiology of prolonged thrombocytopenia is multifactorial and has been associated with stem cell donor relation, infections, adverse events (AEs) related to the medication regimens received, total body irradiation, and immune-mediated responses, such as graft-versus-host disease (GVHD).3,4 Despite the incidence of prolonged thrombocytopenia after HSCT, there are currently no standard guidelines for its management in this patient population, and blood product support is often used. Blood products are used to sustain platelet levels above a specific level, typically 10,000 to 30,000 mm3, when patients have post-HSCT thrombocytopenia.3 Patients often become dependent on these transfusions to maintain an appropriate platelet level, and this comes with a heavy time and financial burden for patients and healthcare institutions. In addition, transfusions are associated with several AEs, such as platelet refractoriness, acute lung and cardiac injury, and infusion-related reactions.3
Eltrombopag is a thrombopoietin receptor agonist that increases platelet counts by activating the human thrombopoietin receptor that has been useful in the setting of post-HSCT thrombocytopenia.5 Thrombopoietin is produced by the liver and helps regulate platelet production by interacting with the c-Mpl receptor on megakaryocytes leading to platelet maturation.6 Although thrombopoietin is largely involved in platelet production, it also has a role in early progenitor cells of other blood lineages.6,7 The thrombopoietin c-Mpl receptor is shown on mature megakaryocytes and platelets, CD34+ cells, pluripotential stem cells, and immature cells of all lineages.6 In animal studies, mice that lacked the thrombopoietin c-Mpl receptor were deficient in hematopoietic stem cells, showing that thrombopoietin is potentially involved in hematopoietic stem-cell homeostasis.7,8 Few human studies have shown that neutrophil and hemoglobin levels are affected, as well as platelets, by treatment with eltrombopag.7,9
Eltrombopag is currently approved for the treatment of aplastic anemia, hepatitis C–associated thrombocytopenia, and immune thrombocytopenia, but evidence is predominantly limited to small, retrospective studies and 1 randomized trial in the setting of post-HSCT thrombocytopenia.5,9-13 Eltrombopag dosing, duration, and response rates for use in this setting have significantly varied among these analyses.9-13 Our study characterizes the efficacy and safety of eltrombopag in the setting of post-HSCT thrombocytopenia and focuses on platelet response rates.
Methods
This single-center, retrospective, observational study was conducted at an academic medical center. The patients who were eligible for study inclusion were aged ≥18 years and had a history of allogeneic HSCT, had post-HSCT thrombocytopenia with platelet counts of <50,000/µL, and began treatment with eltrombopag between January 1, 2017, and August 30, 2021. The study’s exclusion criteria were having an allogeneic transplant for aplastic anemia, having an allogeneic transplant completed at an outside institution, initiating treatment with eltrombopag >4 years after having a transplant, discontinuing treatment with eltrombopag as the result of an inability to swallow, and receiving treatment with eltrombopag for <7 days.
The primary study objective was to characterize the response rate of eltrombopag in patients with thrombocytopenia, which was defined as having a platelet count of ≥50,000/µL in those who had an allogeneic HSCT. The secondary objectives included an evaluation of eltrombopag dose at the time of response; the days to response; the duration of the response; the response rate with eltrombopag in patients who did not receive platelet transfusions in the previous 7 days; the median platelet, white blood cell (WBC), neutrophil, and hemoglobin count change after receiving eltrombopag; the reasons for the discontinuation of treatment with eltrombopag; and the safety events.
The platelet transfusion data were documented throughout the study to characterize the response rates based on the receipt of a platelet transfusion while receiving treatment with eltrombopag as a secondary end point. The laboratory test results were obtained after having an HSCT at baseline, throughout treatment, and 2 to 4 weeks after the completion of eltrombopag treatment. The baseline laboratory test results must have been collected within 7 days before the initiation of treatment with eltrombopag after having an HSCT.
Descriptive statistics were used to analyze the data. Chi-square test and Fisher’s exact test were used to perform the subgroup analyses using IBM SPSS Statistics version 28 (IBM Corporation; Armonk, NY). The study protocol was approved by the University of Kansas Medical Center Institutional Review Board.
Results
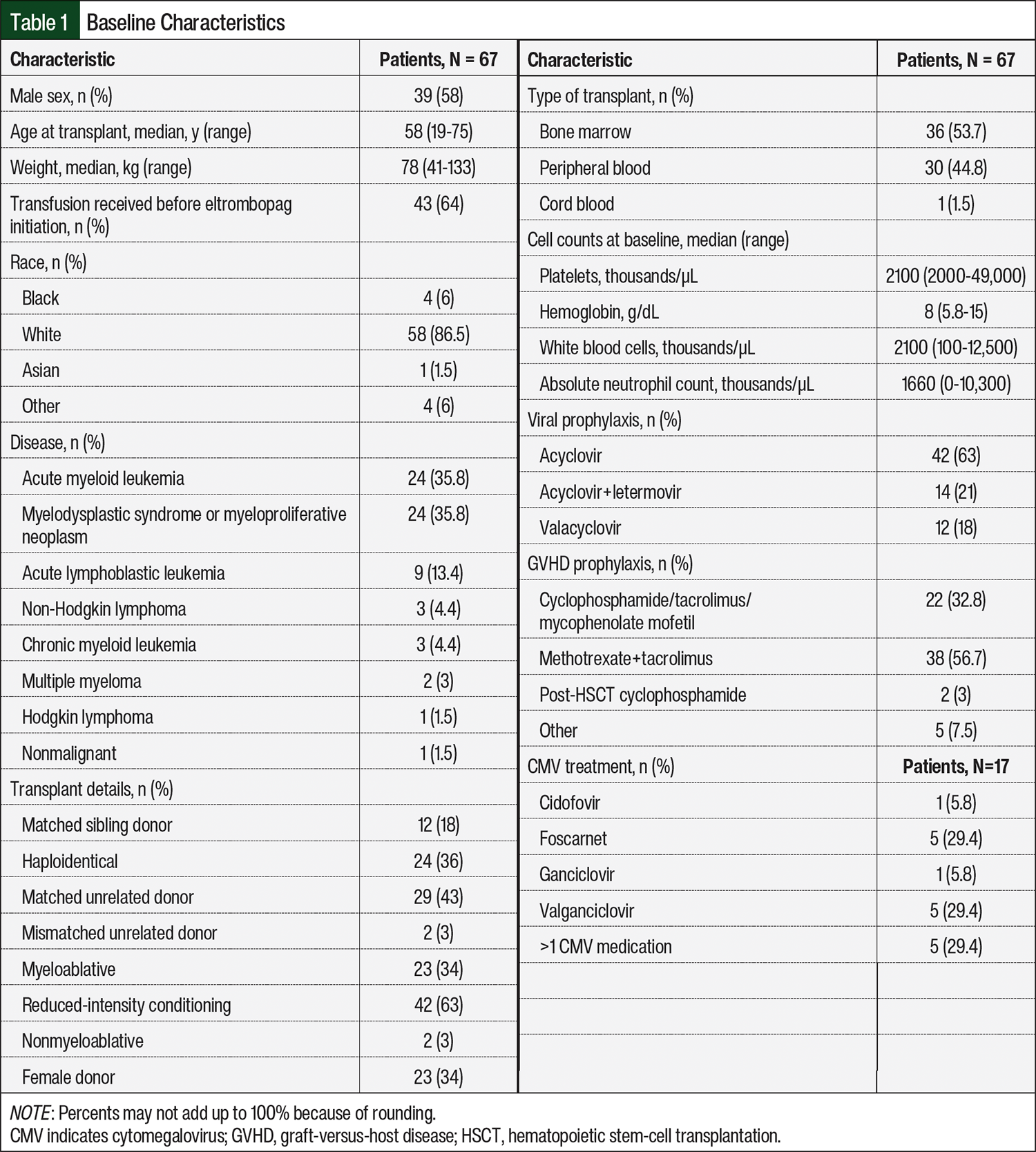
Between January 1, 2017, and August 30, 2021, 74 patients met the inclusion criteria and received eltrombopag for the treatment of posttransplant thrombocytopenia. Of these 74 patients, 7 patients were excluded from the study because they received the transplant at an outside health system (n=1), eltrombopag was initiated >4 years after having a transplant (n=1), they had an inability to swallow eltrombopag at any point during the study (n=3), and their laboratory monitoring was done at an outside health system (n=2). A total of 67 patients were included in the final retrospective analysis (Table 1). There were 39 (58%) men, and the median age at the time of transplant was 58 years (range, 19-75 years). The leading indications for transplant in the 67 patients were acute myeloid leukemia and myelodysplastic syndrome (n=24 [35.8%] each). The source of stem cells in this population was largely bone marrow in 36 (53.7%) patients and peripheral blood in 30 (44.8%) patients, but 1 (1.5%) patient received a cord blood transplant. The conditioning regimens included reduced intensity in 42 (63%) patients, myeloablative in 23 (34%) patients, and nonmyeloablative in 2 (3%) patients.
The median starting dose of eltrombopag was 50 mg (range, 25-150 mg) daily. There was no dosing protocol followed in this study, because eltrombopag was dosed based on the provider’s preference. The doses were titrated in 55 (82%) of the 67 patients, whereas 12 (18%) of the 67 patients received the same dose throughout the duration of their eltrombopag treatments. Notably, the doses were titrated even after the primary outcome of a platelet level of ≥50,000/µL was achieved in 11 (28%) of the 39 patients who achieved a response. The median dose at 1 week was 50 mg (range, 25-150 mg), and the median dose at each time point of 2 weeks, 3 weeks, and 4 weeks was 100 mg (range, 25-150 mg). At 8 and 12 weeks, the median dose of eltrombopag was 150 mg (range, 25-150 mg).
The median time to the start of treatment with eltrombopag was 90 days after having a transplant (range, 25-817 days), and the patients continued receiving treatment with eltrombopag for a median of 136 days (range, 9-1016 days). The median dose of eltrombopag at the time of response was 100 mg (range, 50-150 mg), and the median time to reach a response was 28 days (range, 7-84 days).
Of the 67 patients enrolled in the study, 39 (58%) achieved a platelet level of ≥50,000/µL after receiving eltrombopag. In all, 32 (48%) of these 67 patients were transfusion independent in the 7 days before achieving a platelet response with eltrombopag. After achieving a platelet response, 28 (72%) of the 39 responders sustained a platelet level of ≥50,000/µL 2 to 4 weeks after treatment with eltrombopag was discontinued. Of the 39 responders, 3 (8%) did not sustain platelets >50,000/µL and the other 9 (23%) did not have follow-up laboratory testing within 2 to 4 weeks after the response as a result of death, follow-up outside of the health system, follow-up outside of the designated time window, or receiving hospice care.
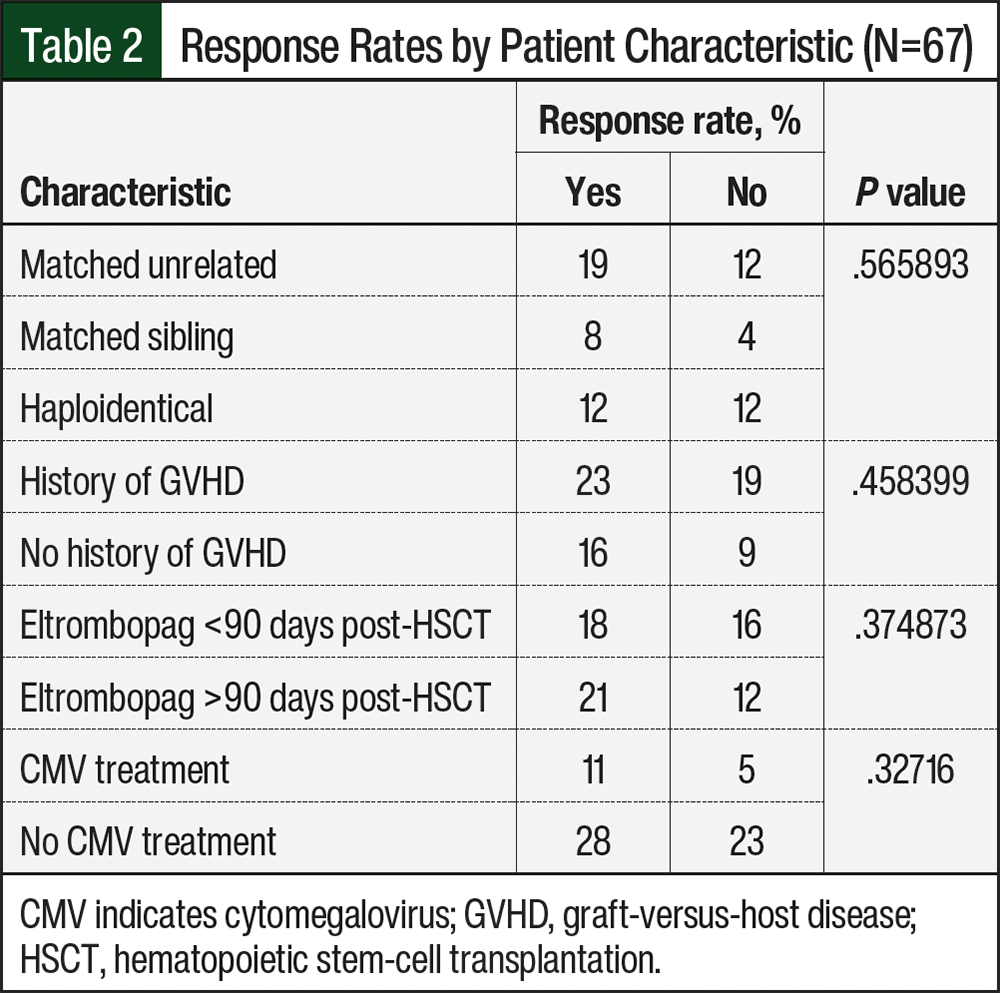
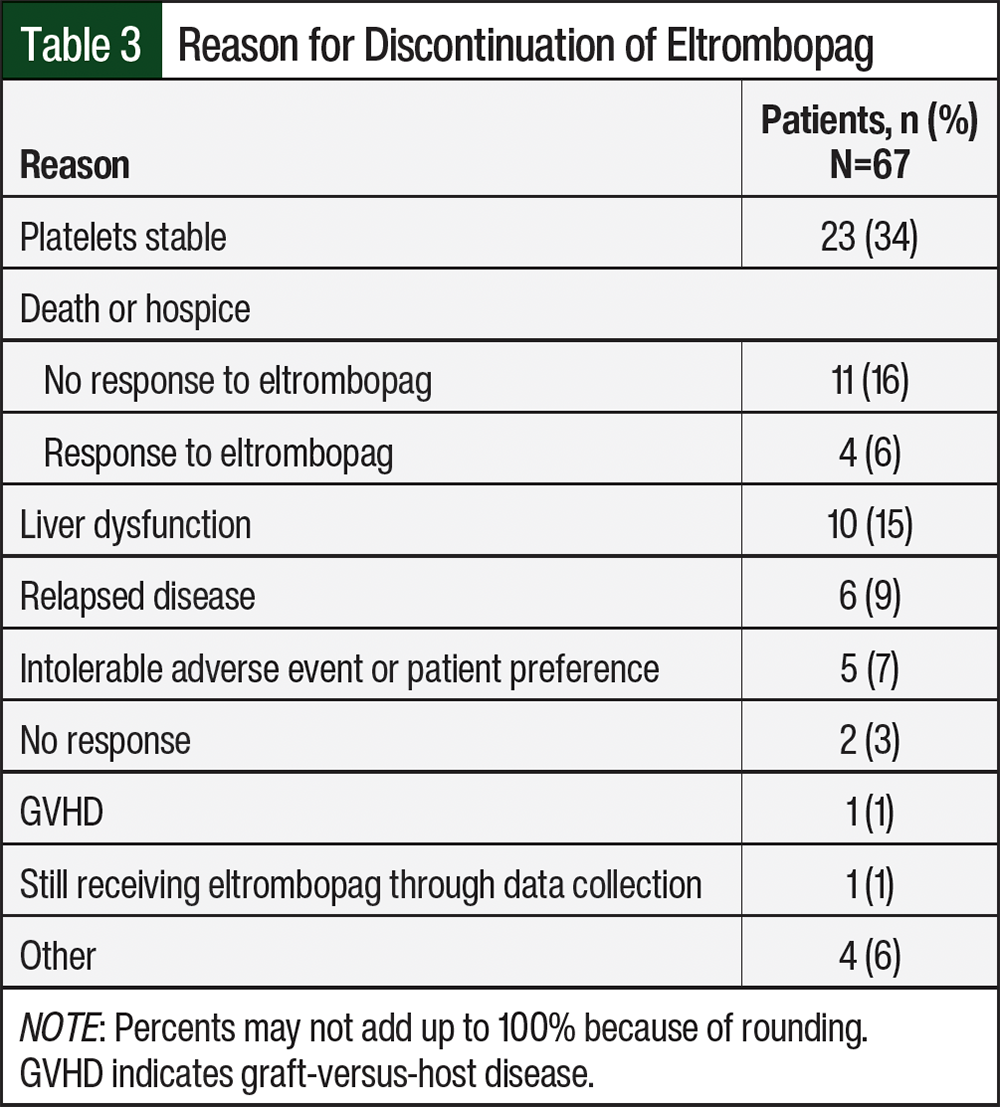
There were no statistical differences in the subgroup analysis comparing the response rates with GVHD history, the initiation of treatment with eltrombopag before or after 3 months in the posttransplant period, cytomegalovirus treatment, or the type of transplant (Table 2). In the 67 patients, the most common reasons for the discontinuation of treatment with eltrombopag included platelets being deemed stable by the provider in 23 (34%) patients, death or hospice in 15 (22%) patients, and hyperbilirubinemia in 10 (15%) patients (Table 3).
Grade ≥1 hyperbilirubinemia, which is defined as total bilirubin 1.5 times the upper limit of normal or 1.95 mg/dL, occurred in 25 (37%) of the 67 patients who were receiving eltrombopag. There were no cases of venous thromboembolism. A total of 5 (7%) patients discontinued treatment with eltrombopag as a result of intolerance or preference, with the only documented intolerable AE being headache.
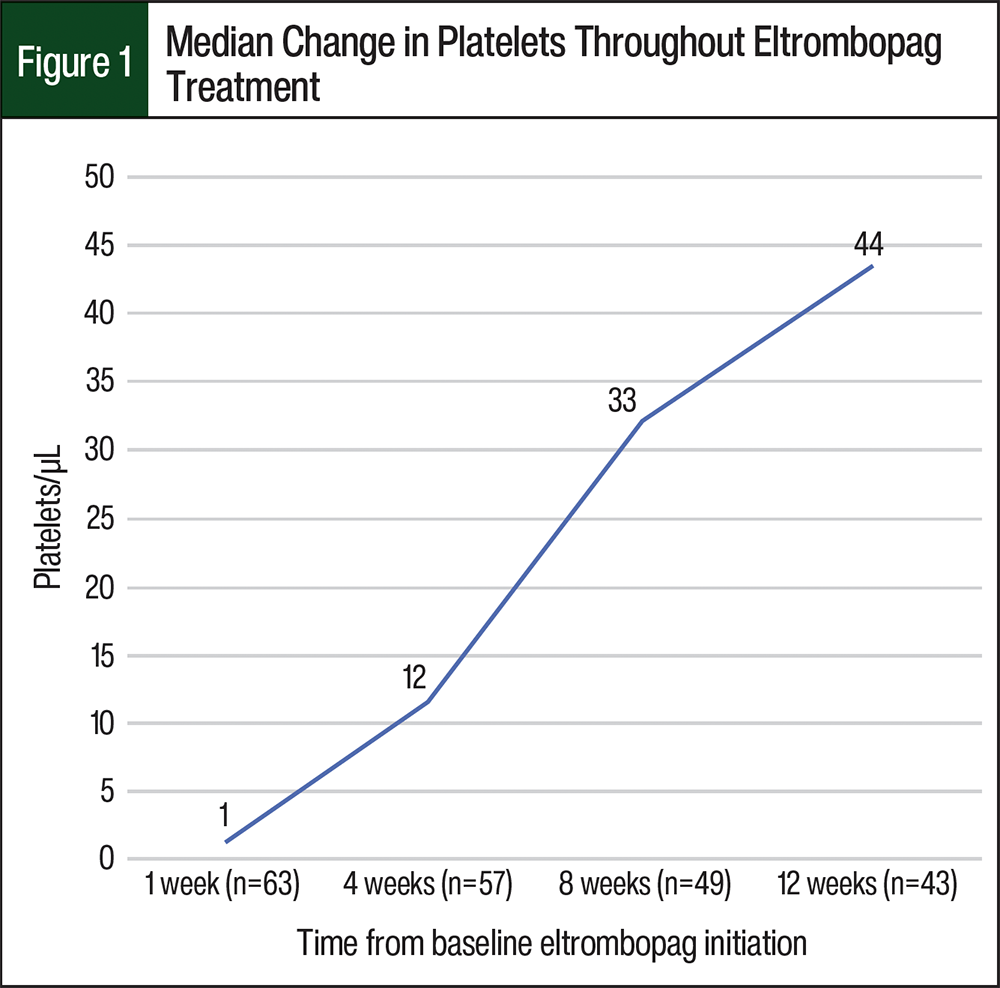
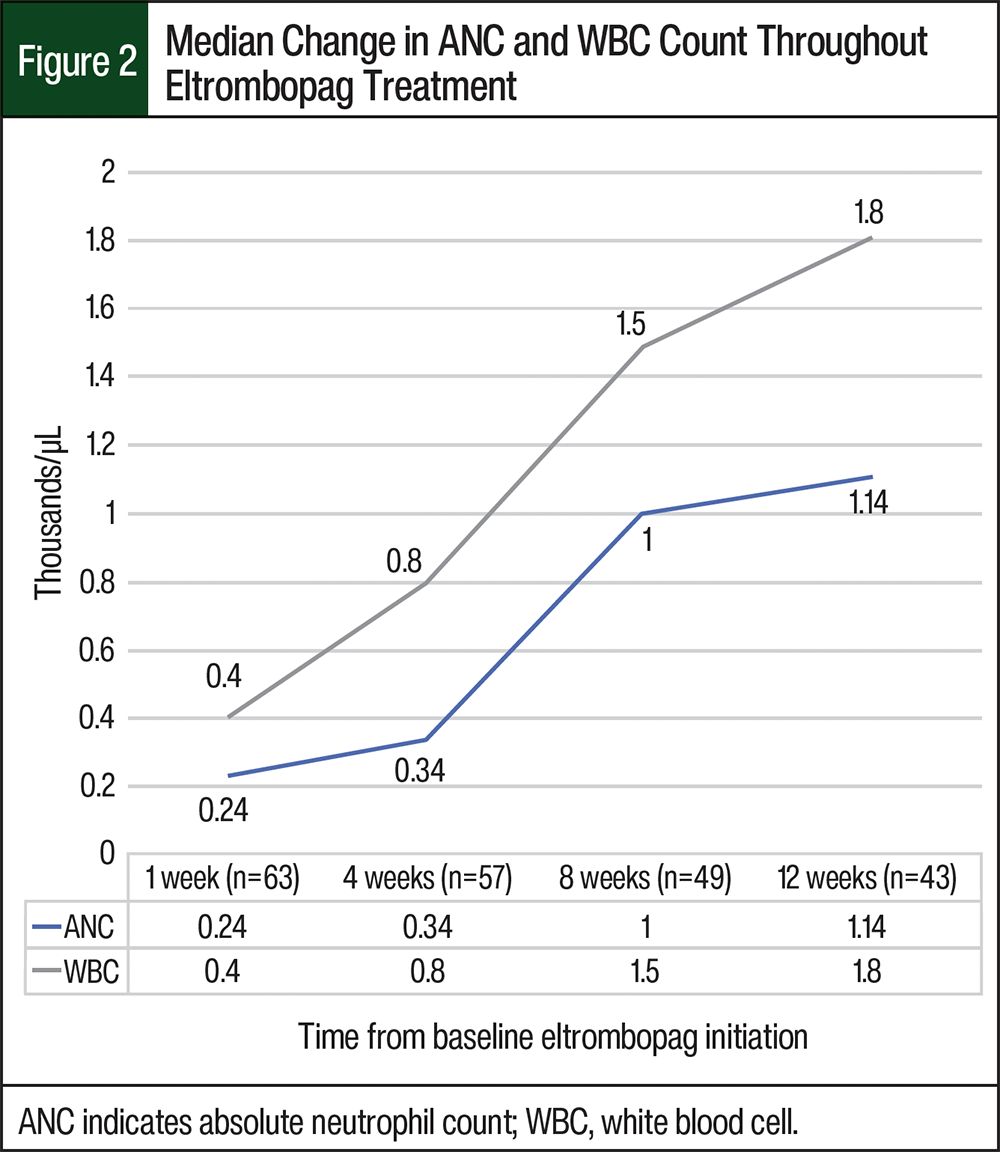
The cell counts at baseline are shown in Table 1. A total of 66 (98%) patients had anemia (hemoglobin <12 g/dL in female patients and <13.5 mg/dL in male patients) before receiving eltrombopag treatment, and 32 (48%) of the 67 patients had neutropenia (absolute neutrophil count [ANC] ≤1500 cells/µL). All patients at baseline had thrombocytopenia with platelets of <50,000/µL and a median platelet count of 21,000/µL (range, 2000-49,000/µL). Throughout treatment with eltrombopag, blood counts were observed after 1 week (n=63), 4 weeks (n=57), 8 weeks (n=49), and 12 weeks (n=43); the median changes in platelet, ANC, and WBC counts from baseline are shown in Figure 1 and Figure 2.
Notably, the median increases in blood counts from baseline to 12 weeks of receiving eltrombopag were 0.9 mg/dL for hemoglobin, 1140 cells/µL for ANC, and 1800 cells/µL for the WBC count. After the discontinuation of treatment with eltrombopag, platelets continued to increase. The median platelet count 2 to 4 weeks after the discontinuation of treatment with eltrombopag increased from the median platelet count of 65,000/µL at 12 weeks (10,000-280,000/µL) to 105,000/µL (4000-344,000/µL).
Discussion
To our knowledge, this is one of the largest studies to report outcomes associated with eltrombopag treatment for postallogeneic transplant thrombocytopenia. Our results support treatment with eltrombopag after a transplant, according to this study’s results of 39 (58%) responders of the 67 total study patients who received a median dose of eltrombopag of 100 mg (range, 50-150 mg) and a median time to response of 28 days (range, 7-84 days). Of the 39 responders, 28 (72%) were able to sustain the platelet response 2 to 4 weeks after the discontinuation of treatment with eltrombopag. This study adds to the literature available on treatment with eltrombopag for post-HSCT thrombocytopenia, expanding beyond the limited data to help characterize the response rates, duration of treatment, median time to platelet response, and median change in platelets in a population of patients who had allogeneic stem-cell transplant.
At the time of this retrospective analysis, the results of the only randomized trial that evaluated posttransplant treatment with eltrombopag showed a response in 9 (21%) of the 42 patients who received eltrombopag.10 This response rate is much lower than the rate in our retrospective analysis (58% including transfusions, 48% for the transfusion-independent rate) and in previous retrospective data that have reported highly variable response rates ranging from 60% to 75%.9,11-13 Although variable, it is clear that eltrombopag induces platelet creation and transfusion independence, and previous retrospective data have identified positive predictors for response including HLA-matched donors, a CD34+ dose at transplant >4×106 kg, and eltrombopag initiation ≥90 days after a transplant.9 Although not statistically significant, a similar difference was noted in our subgroup analysis, in which 21 (64%) of the 33 patients who started treatment with eltrombopag ≥90 days after a transplant achieved a response compared with only 18 (53%) of 34 patients who started treatment with eltrombopag <90 days after a transplant achieved a response.
At baseline, many of the patients in this study had decreased neutrophils and hemoglobin. Thrombopoietin theoretically has an impact on these cell lineages, given the presence of the thrombopoietin c-Mpl receptor on megakaryocytes, CD34+ cells, pluripotential stem cells, and other immature cells in all lineages, but this impact is not well-defined in the literature.6-8 In our study, the ANC increased by a median of 1140 cells/µL and the hemoglobin increased by a median of 0.9 g/dL after 12 weeks of receiving treatment with eltrombopag. These data support the potential for eltrombopag to affect other cell lineages, but to a minimal extent, and should be validated by other large, randomized trials.
Similar to previous literature, eltrombopag was well-tolerated among patients in this study without any incidence of venous thromboembolism, and only 5 (7%) of the 67 patients discontinued therapy as a result of personal preference or headache.11-13 The known AE of grade ≥1 hyperbilirubinemia was observed in 25 (37%) of the 67 patients included and led to the discontinuation of treatment in 10 (15%) of the 67 patients. Previous trials have shown that grade 3 hyperbilirubinemia occurred in 14% of patients10 and grade ≥2 hyperbilirubinemia occurred in 13.2% of patients.12 The increased rate of hyperbilirubinemia in this study may be attributed to the inclusion of grade ≥1 hyperbilirubinemia as well as confounding factors, including the presence of GVHD or receiving concomitant medications.
To be included in this study, patients had to receive eltrombopag for at least 7 days, and their medication history was used to determine eligibility for the study. Notably, 19 patients were not included in this analysis because treatment with eltrombopag was never started as a result of cost. This alone highlights the unfortunate financial barrier to treatment with eltrombopag in this setting and the financial toxicity that patients can have while receiving treatment with eltrombopag. Although cost was not analyzed in this study, it is unclear if the financial burden outweighs the benefits in all patients who qualify for treatment with eltrombopag after a transplant.
Limitations
This study has limitations. The limitations include the retrospective nature of this study, which impacted the amount of data points at each time point throughout treatment with eltrombopag. Because this was a single-site analysis and the data were collected before the institutional implication of posttransplant treatment with cyclophosphamide, these results are only applicable to the conditioning and GVHD regimens that were used. In addition, 16 (24%) of the 67 patients received treatment throughout the study with cytomegalovirus, some of which included ganciclovir and valganciclovir which are known to impact cell counts. The impact this may have had on the study was assessed using subgroup analysis, which showed no statistical differences between responders and nonresponders regarding cytomegalovirus treatment.
Conclusion
Patients with posttransplant allogeneic thrombocytopenia may benefit from treatment with eltrombopag to achieve a platelet level of ≥50,000/µL, but this response may take several weeks to reach. Future large, prospective studies are needed to evaluate the impact and risks versus benefits of eltrombopag treatment for patients with posttransplant thrombocytopenia, because the current data are predominantly limited to retrospective studies.
Acknowledgments
SB performed the research; SB, MN, DB, and ZM contributed to the data analysis and interpretation; and DG served as project statistician. All authors contributed to the review, provided comments on this manuscript, and approved the final version of this article.
Author Disclosure Statement
Dr Nelson is on the Advisory Board and Unbranded Speaker Bureau of Janssen Biotech; Dr Blocker, Dr Mahmoudjafari, Dr Cascone, Dr Grauer, and Dr Beckman have no conflicts of interest to report.
References
- Bolwell B, Pohlman B, Sobecks R, et al. Prognostic importance of the platelet count 100 days post allogeneic bone marrow transplant. Bone Marrow Transplant. 2004;33:419-423.
- Yamazaki R, Kuwana M, Mori T, et al. Prolonged thrombocytopenia after allogeneic hematopoietic stem cell transplantation: associations with impaired platelet production and increased platelet turnover. Bone Marrow Transplant. 2006;38:377-384.
- Bento L, Canaro M, Bastida JM, Sampol A. Thrombocytopenia and therapeutic strategies after allogeneic hematopoietic stem cell transplantation. J Clin Med. 2022;11:1364.
- Dominietto A, Raiola AM, van Lint MT, et al. Factors influencing haematological recovery after allogeneic haemopoietic stem cell transplants: graft-versus-host disease, donor type, cytomegalovirus infections and cell dose. Br J Haematol. 2001;112:219-227.
- Promacta (eltrombopag) tablets, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; March 2023. Accessed September 1, 2023. www.novartis.com/us-en/sites/novartis_us/files/promacta.pdf
- Mahat U, Rotz SJ, Hanna R. Use of thrombopoietin receptor agonists in prolonged thrombocytopenia after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2020;26:e65-e73.
- Desmond R, Townsley DM, Dumitriu B, et al. Eltrombopag restores trilineage hematopoiesis in refractory severe aplastic anemia that can be sustained on discontinuation of drug. Blood. 2014;123:1818-1825.
- Alexander WS, Roberts AW, Nicola NA, et al. Deficiencies in progenitor cells of multiple hematopoietic lineages and defective megakaryocytopoiesis in mice lacking the thrombopoietic receptor c-Mpl. Blood. 1996;87:2162-2170.
- Giammarco S, Sica S, Chiusolo P, et al. Eltrombopag for the treatment of poor graft function following allogeneic stem cell transplant: a retrospective multicenter study. Int J Hematol. 2021;114:228-234.
- Ahmed S, Bashir Q, Bassett R, et al. Eltrombopag for post-transplantation thrombocytopenia: results of phase II randomized, double-blind, placebo-controlled trial. Transplant Cell Ther. 2021;27:430.e1-430.e7.
- Bento L, Bastida JM, García-Cadenas I, et al. Thrombopoietin receptor agonists for severe thrombocytopenia after allogeneic stem cell transplantation: experience of the Spanish Group of Hematopoietic Stem Cell Transplant. Biol Blood Marrow Transplant. 2019;25:1825-1831.
- Fu H, Zhang, Xiaohui, Han T, et al. Eltrombopag is an effective and safe therapy for refractory thrombocytopenia after haploidentical hematopoietic stem cell transplantation. Bone Marrow Transplant. 2019;54:1310-1318.
- Yan F, Lu N, Gu Z, et al. Eltrombopag in the treatment of patients with persistent thrombocytopenia after haploidentical peripheral blood stem cell transplantation: a single-center experience. Ann Hematol. 2022;101:397-408.