Connecting Science to Practice
Multiple myeloma is a blood cancer with an increased risk
for venous thromboembolism (VTE). The authors used the
IMPEDE score to stratify VTE risk for patients with multiple
myeloma receiving active treatment and evaluated the
safety and efficacy of the prescribed VTE prophylaxis, including
the National Comprehensive Cancer Network
(NCCN)-directed VTE prophylaxis (representing optimal
prophylaxis). We found a lower VTE incidence in those
receiving optimal VTE prophylaxis compared with patients
receiving suboptimal prophylaxis (4% vs 6.7%) and a minimal
bleeding risk with chemical VTE prophylaxis (0.8%).
Our study showcases the need for NCCN-directed VTE
prophylaxis in vulnerable patients based on IMPEDE scoring.
We conclude that pharmacist-led implementation
should be studied to determine an appropriate workflow.
Cancer is an independent risk factor for venous thromboembolism (VTE) events, with an incidence of 5% to 20% of patients having a breakthrough VTE.1 The risk for VTE is even higher in patients with multiple myeloma, with the highest risk during the initial 6 months after diagnosis.2,3 Furthermore, the pharmacotherapy agents for multiple myeloma, including immunomodulatory drugs (IMiDs) combined with chemotherapy or steroids, further increase the risk for VTE.4
Several VTE risk tools and calculators have been established to assess the risk for breakthrough VTE events in patients with multiple myeloma, of which the SAVED and IMPEDE scores are the most used in clinical practice.5 The SAVED score was created to stratify the risk for IMiD-associated VTE events in patients with newly diagnosed multiple myeloma.6 The components of the SAVED score include surgery in the past 90 days, Asian ethnicity, VTE history, age ≥80 years, and monthly use of dexamethasone. The SAVED score was validated in more than 500 patients with newly diagnosed multiple myeloma from 2010 to 2019 who received treatment at the Cleveland Clinic. The study confirmed that the SAVED score could adequately stratify the risk for VTE occurrence in patients with newly diagnosed multiple myeloma who were receiving IMiD-based treatment (C-statistic=.74).6
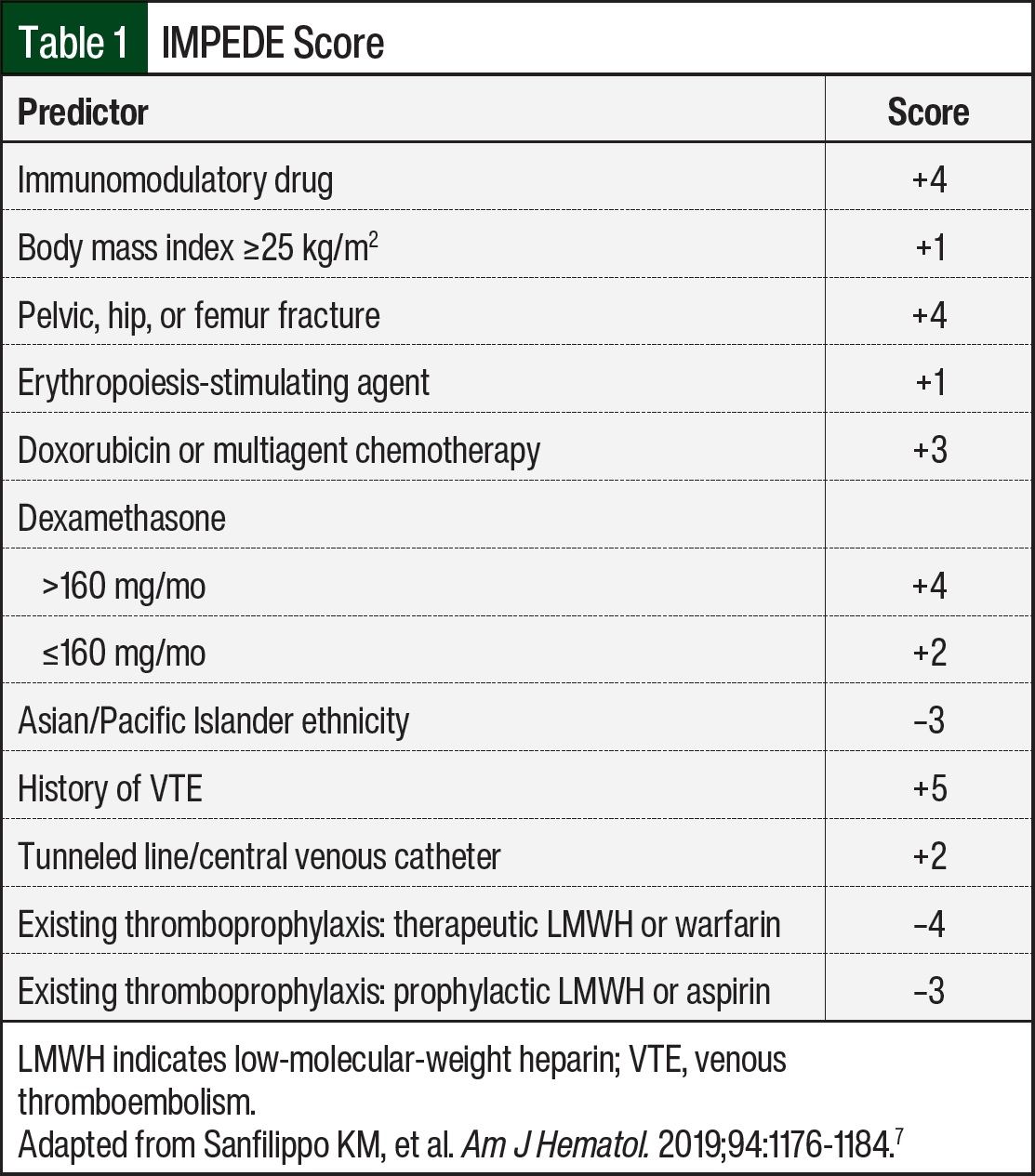
The IMPEDE score is more inclusive than the SAVED score because IMPEDE is designed for all patients with newly diagnosed multiple myeloma. The components of the IMPEDE score include IMiDs, body mass index, fractures, central venous catheter, therapeutic low-molecular-weight heparin (LMWH) or warfarin, prophylactic LMWH or aspirin, ethnicity, steroid use, history of VTE, doxorubicin, and erythropoiesis-stimulating agent (Table 1).7 The IMPEDE score was validated in 575 patients with newly diagnosed multiple myeloma regardless of IMiD use (C-statistic=.68).5
The National Comprehensive Cancer Network (NCCN) provides clinical practice guidelines for VTE prophylaxis in patients with multiple myeloma.5 The NCCN recommends using either the IMPEDE or SAVED scoring system to determine the type of VTE prophylaxis that patients with multiple myeloma should receive. Patients with an IMPEDE score of ≤3 or a SAVED score of <2 should receive aspirin 81 to 325 mg daily, whereas patients with an IMPEDE score of ≥4 or a SAVED score of ≥2 should receive a prophylactic dose of LMWH, rivaroxaban, apixaban, fondaparinux, or warfarin (Table 2).5
Research has established several validated VTE risk stratification tools for patients with multiple myeloma, and the NCCN has provided recommended VTE prophylaxis based on a patient’s risk stratification.5-8 Gaps in the literature exist regarding validating the efficacy and safety of the NCCN guideline recommendations for VTE prophylaxis in patients with multiple myeloma. These NCCN-directed VTE prophylaxis recommendations should be assessed in terms of their effectiveness in the prevention of breakthrough VTE events and the incidence of bleeding. The purpose of this study is to use the IMPEDE score to stratify the VTE risk of patients with multiple myeloma who are actively receiving treatment and to evaluate for the incidence of breakthrough VTE and potential gaps within the initiation of VTE prophylaxis.
Methods
This single-center, retrospective quality improvement study was performed at a 950-bed community, not-for-profit hospital and an affiliated outpatient cancer center. Between January 2023 and June 2023, a random sample of patients with an International Classification of Diseases, Tenth Revision code specific for multiple myeloma (C90.0) was used. The study’s inclusion criteria were patients aged ≥18 years with at least 1 treatment cycle for multiple myeloma. Patients were excluded from the study if they had not received treatment for multiple myeloma during this time frame or if they had insufficient follow-up with the outpatient cancer center.
Electronic health records (EHRs) were analyzed to collect the patient’s sex and age, IMPEDE score components (Table 1), VTE events, chemical VTE prophylaxis, and bleeding events. The medication dosage and frequency for chemical VTE prophylaxis were recorded and were used to assess if guideline-directed VTE prophylaxis was used.
The primary study outcome was to evaluate the incidence of breakthrough VTE in patients receiving active treatment for multiple myeloma within 6 months of study inclusion. This 6-month follow-up coincided with the highest risk for breakthrough VTE events in patients with newly diagnosed multiple myeloma and provided a reasonable lookback period for all other patients with multiple myeloma. The secondary study outcomes include the incidence of bleeding within 6 months of study inclusion and the rate of compliance with prescribing guideline-directed therapy for VTE prophylaxis based on the IMPEDE score and the NCCN guidelines. The terminology used to represent optimal compliance was when the VTE prophylaxis coincided with the NCCN’s guideline recommendations based on the IMPEDE score or when the documented bleeding risk prevented optimal VTE prophylaxis. The terminology for suboptimal compliance was when the VTE prophylaxis did not coincide with the NCCN’s guideline recommendations based on the IMPEDE score.
Statistical Analysis
The patients’ baseline characteristics, including age and total IMPEDE score, were summarized by interquartile ranges, and they were analyzed using an independent sample t-test for parametric variables and the Mann–Whitney U test for nonparametric variables. Percentages and numbers summarized the primary outcome, secondary outcomes, and other baseline characteristics, and a chi-square test analyzed them. The criterion for statistical importance was a P value of ≤.05. Statistics for this quality improvement project were used descriptively, and no power calculation was performed to determine a sample size. The statistics should be interpreted accordingly.
Results
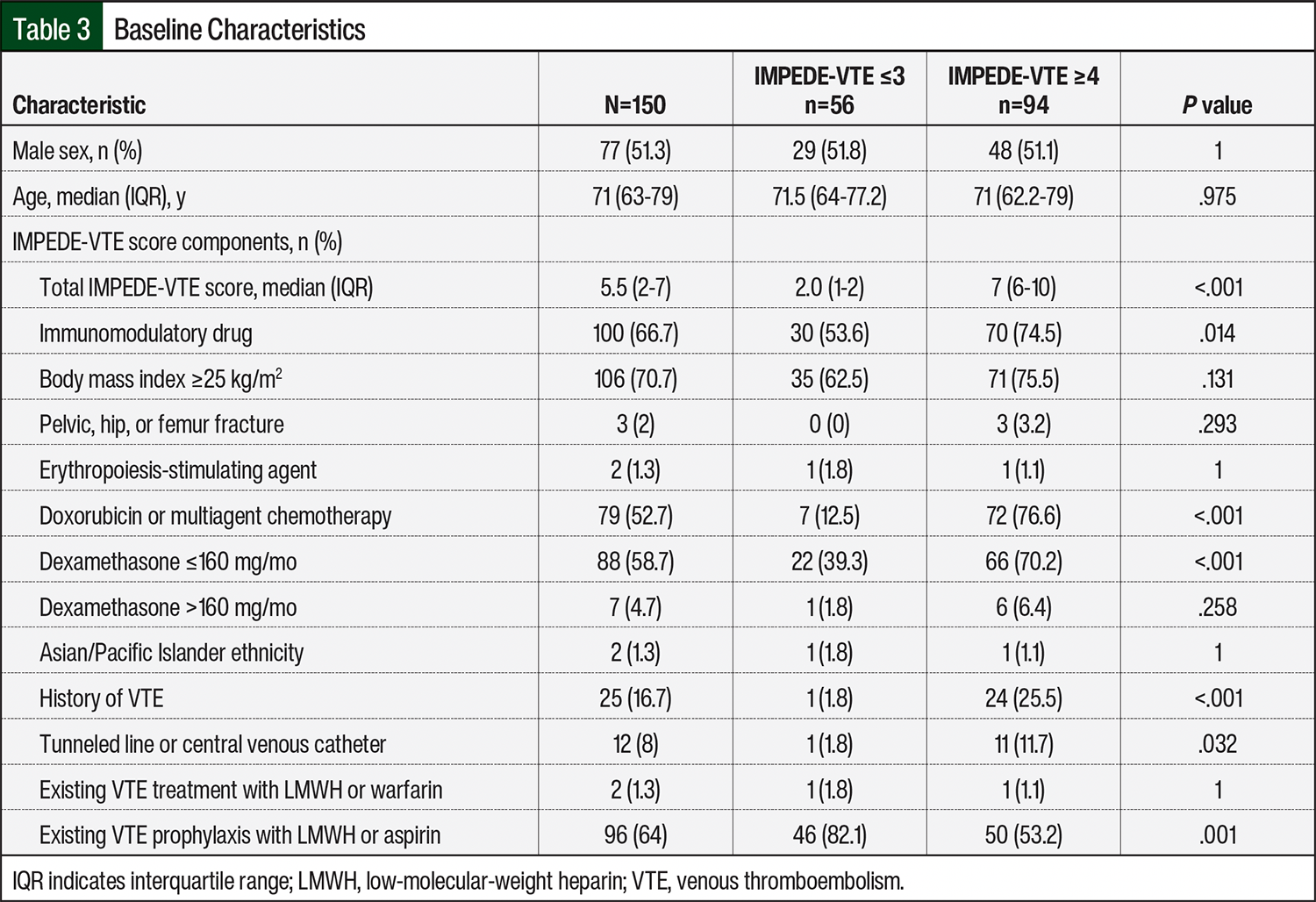
After randomization, 209 patients were assessed for study inclusion. In all, 31 patients were excluded from the study because they were not actively receiving treatment for multiple myeloma, and 28 patients were excluded as a result of insufficient follow-up at the outpatient cancer center. Therefore, 150 patients met the study’s inclusion criteria, with 94 patients having an IMPEDE score of ≥4 (high risk) and 56 patients having an IMPEDE score of ≤3 (low risk).
There were several distinct differences within the baseline characteristics of the high-risk and low-risk IMPEDE score groups (Table 3). The pertinent differences included increased occurrences of the following IMPEDE score components in the high-risk group compared with the low-risk group: IMiDs (74.5% vs 53.6%, respectively; P=.014), receiving doxorubicin or multiagent chemotherapy (76.6% vs 12.5%, respectively; P<.001), receiving dexamethasone at a dose of ≤160 mg monthly (70.2% vs 39.3%, respectively; P<.001), having a history of VTE (25.5% vs 1.8%, respectively; P<.001), and having a tunneled line or central venous catheter (11.7% vs 1.8%, respectively; P=.032). Another pertinent difference was an increased occurrence of existing VTE prophylaxis with LMWH or aspirin in the low-risk group compared with the high-risk group (82.1% vs 53.2%, respectively; P=.001).
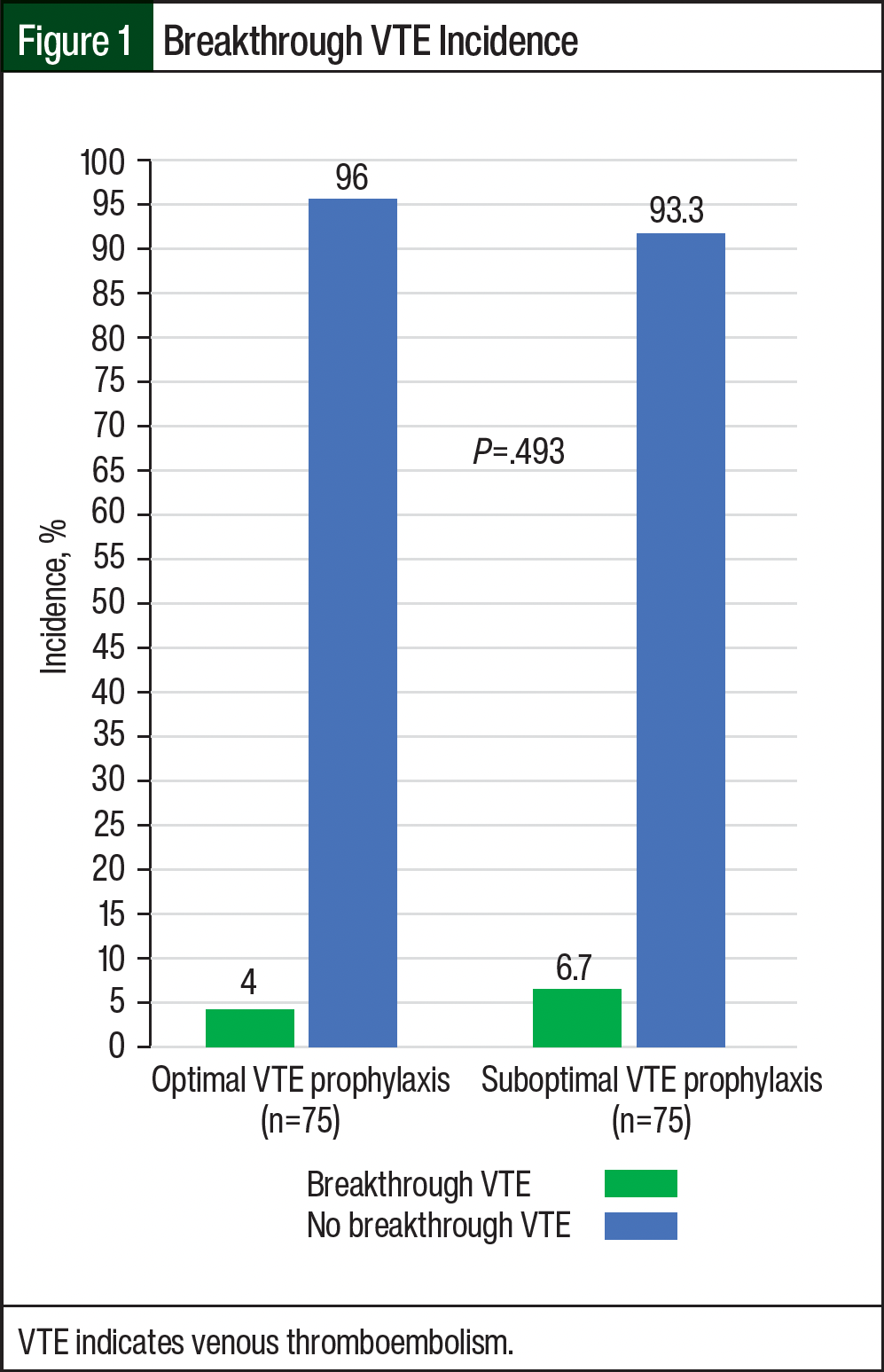
For the primary outcome, 8 breakthrough VTE events occurred (Figure 1). There were 3 VTE events in the 75 patients with optimal VTE prophylaxis and 5 VTE events in the 75 patients with suboptimal VTE prophylaxis (4% vs 6.7%, respectively; P=.493; absolute risk reduction, 2.7%). Further analysis revealed that 7 of the 8 patients with breakthrough VTE events received full-dose anticoagulation, and 1 of the 8 patients with breakthrough VTE events received prophylactic dose anticoagulation. There were 7 deep vein thrombosis (DVT) events and 2 pulmonary embolisms (PEs); 1 of these patients with optimal VTE prophylaxis had a PE and DVT simultaneously, which was counted as 1 VTE event. The location of the VTE events included the lower extremities, chest, and neck. The analysis also showed that 3 of the 8 breakthrough VTE events occurred in patients with no chemical prophylaxis, and 2 of the 8 breakthrough events occurred in patients with newly diagnosed multiple myeloma. Of the 3 breakthrough VTE events that occurred in patients who did not receive chemical prophylaxis, 2 occurred in high-risk patients and 1 in a low-risk patient.
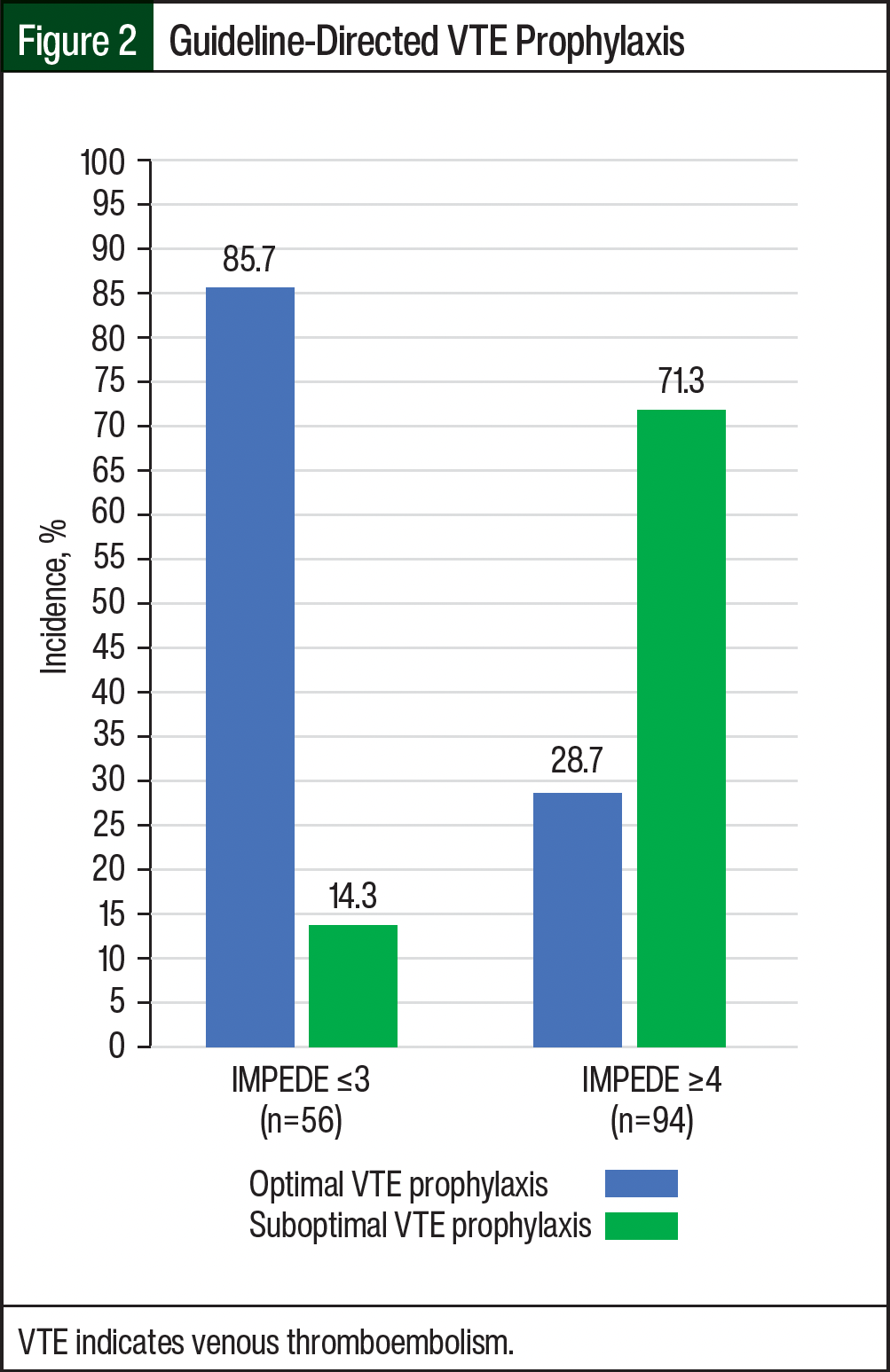
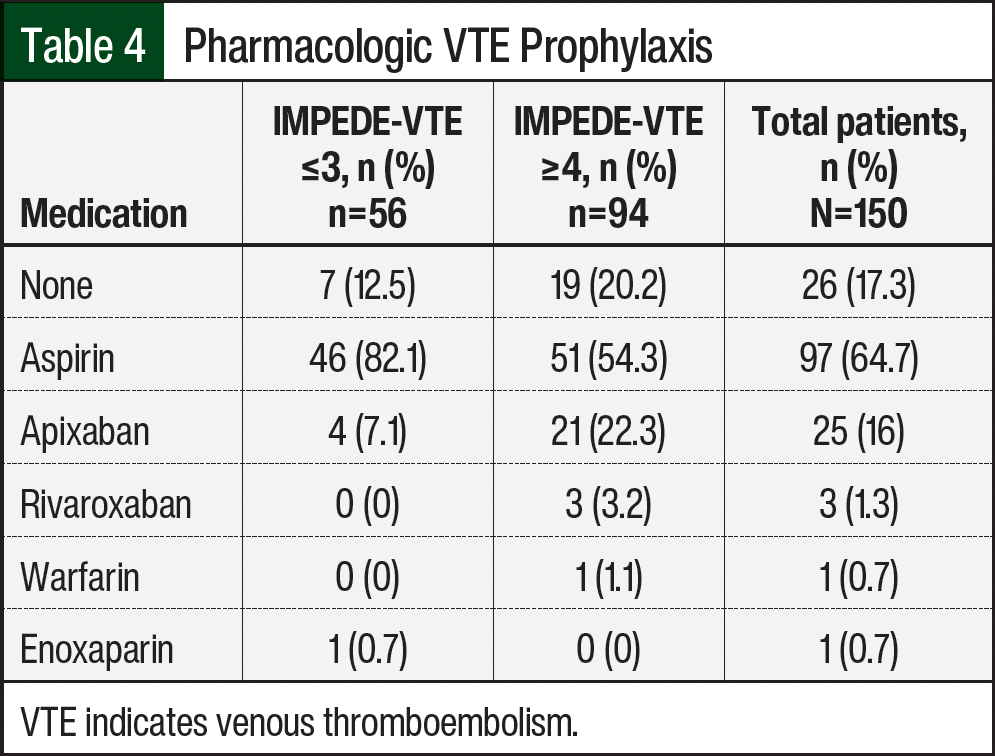
For the secondary outcomes, there was 1 bleeding event (hematuria) in the 124 patients who received chemical VTE prophylaxis (Figure 2). For compliance with optimal VTE prophylaxis based on the NCCN’s guideline recommendations and IMPEDE score, 75 (50%) of 150 patients had optimal VTE prophylaxis (Figure 2). When stratified based on VTE risk, the low-risk group had 48 (85.7%) of 56 patients with optimal VTE prophylaxis and the high-risk group had 27 (28.7%) of 94 patients with optimal VTE prophylaxis. Further analysis showed that 7 (12.5%) of 56 low-risk patients and 19 (20.2%) of 94 high-risk patients did not receive chemical VTE prophylaxis. The most often prescribed chemical VTE prophylaxis for low-risk and high-risk patients was aspirin, which was received by 46 (82.1%) of 56 low-risk patients and by 51 (54.3%) of 94 high-risk patients (Table 4).
Discussion
Two retrospective studies whose primary outcome was to validate the IMPEDE score analyzed VTE prophylaxis efficacy as a secondary finding.9,10 Bravo-Perez and colleagues analyzed the IMPEDE scores of 438 patients with multiple myeloma in Spain from 1991 to 2020.9 The study externally validated the IMPEDE score and showed decreased breakthrough VTE events with heparin compared with aspirin for VTE prophylaxis (6.4% vs 13%, respectively; hazard ratio [HR], 0.15; P=.001). The investigators concluded that the type of VTE prophylaxis might be a predictive value for VTE in patients with multiple myeloma.9
Bao and colleagues analyzed the IMPEDE scores of 405 patients with newly diagnosed multiple myeloma from 2013 to 2022.10 The study validated the IMPEDE score (C-statistic=.74) and proposed the increased efficacy of LMWH versus aspirin for VTE prophylaxis in this population. The incidence of breakthrough VTE events in this study was lower in the LMWH group than in the aspirin group (4.5% vs 16.3%, respectively; HR, 0.19; P=.009).10 These studies did not correlate VTE prophylaxis with the IMPEDE score, but they indicated that aspirin might be less effective than LMWH and heparin for VTE prevention in patients with multiple myeloma.9,10 This is the first study to use the IMPEDE score and the NCCN’s VTE prophylaxis recommendations for multiple myeloma to assess for appropriate VTE prophylaxis, breakthrough VTE events, and bleeding episodes.
While examining guideline-directed VTE prophylaxis based on the IMPEDE score and the NCCN’s recommendations, the patients in our study had an elevated incidence of breakthrough VTE events in patients with suboptimal VTE prophylaxis compared with optimal (6.7% vs 4%, respectively). We believe the incidence of breakthrough VTE events was higher in the patients with suboptimal VTE prophylaxis because of the increased use of aspirin in our patients with a high-risk IMPEDE score (54.3%). Aspirin would not be the appropriate VTE prophylaxis for high-risk patients unless there was a significant risk for bleeding. Therefore, these findings coincide with existing literature.9,10 Aspirin has limited efficacy in preventing breakthrough VTE events and should not be used in high-risk patients with multiple myeloma.9,10
Trends for breakthrough VTE events were identified in our study. The patients who did not receive chemical VTE prophylaxis had the highest risk for breakthrough VTE events. In all, 14.3% of the low-risk patients and 11.5% of the high-risk patients who did not receive chemical VTE prophylaxis had a breakthrough event. Also, 2 (25%) of 8 newly diagnosed patients with multiple myeloma who had breakthrough VTE events received suboptimal prophylaxis. This aligns with the findings of Bradbury and colleagues, who analyzed the incidence of thrombosis in the randomized, controlled Myeloma IX and Myeloma XI trials.3 The trials’ results showed that the first 6 months of treatment for multiple myeloma was the time frame in which most breakthrough VTE events occurred.3 These trends highlight the importance of using the IMPEDE score and the NCCN’s recommendations for VTE prophylaxis in patients with multiple myeloma. Patients should receive VTE prophylaxis unless they have had significant bleeding events or if they have a high bleeding risk. During our data analysis, none of the patients had documentation stating that they qualified for withholding prophylaxis.
A significant concern with chemical VTE prophylaxis selection is the risk for bleeding. Many oncologists at our institution are wary about starting medications other than aspirin because of the concern for bleeding events. One retrospective study by Piedra and colleagues analyzed the efficacy and safety of rivaroxaban in patients with multiple myeloma.11 The study showed that rivaroxaban is an effective anticoagulant for the prevention of VTE events (VTE incidence, 4.8%) with a bleeding incidence of 1%.11 Our study’s results aligned with these findings, with a bleeding incidence of 0.8% in all patients who received chemical prophylaxis. The one minor bleeding event was gross hematuria in a patient who received aspirin. Therefore, there was no incidence of bleeding after receiving direct oral anticoagulants. This finding supports the use of prophylactic doses of direct oral anticoagulants in high-risk patients with multiple myeloma.
Another risk stratification tool for patients with multiple myeloma, which was created by Chakraborty and colleagues, is the PRISM score.12 The components of the PRISM score include a history of VTE events, abnormal metaphase cytogenetics, surgery within the past 90 days, Black race, and receiving an IMiD during induction therapy. Their initial validation included 783 patients at the Cleveland Clinic from 2008 to 2018. Chakraborty and colleagues were able to provide initial validation (C-statistic=.622), but their results are still pending external validation.12 In our opinion, the IMPEDE score is currently the most suitable VTE scoring tool for patients with multiple myeloma.
This study has several strengths. Data collection was consistent with 1 investigator. A comprehensive chart review was performed to assess the prevention of VTE. Most of the study’s patients are Hispanic, which is an understudied population. The patients’ documented bleeding risk was taken into consideration when categorizing for optimal or suboptimal VTE prophylaxis.
Limitations
The study has several limitations, including inconsistency with the EHR documentation, the assumption of patient adherence to prescribed thromboprophylactic medications, the calculation of 1 initial IMPEDE score per patient, and the inclusion of only outpatient thromboprophylactic medications. Further, it must be noted that the IMPEDE score’s component doxorubicin or multiagent chemotherapy (Table 1) was an adaptation of Sanfilippo and colleagues’ original scoring tool.7 The NCCN made this adaptation,5 and we used it for data collection and interpreted multiagent chemotherapy as receiving >2 antineoplastic drugs for the treatment of multiple myeloma.
Conclusion
This study’s results show that the risk for breakthrough VTE outweighed the risk for bleeding associated with the use of antithrombotic therapy in patients with multiple myeloma. The results also show that our most vulnerable patients for VTE events are newly diagnosed patients and patients who have not received any form of VTE prophylaxis. Identifying the vulnerable patient populations and showcasing the limited bleeding risk with VTE prophylaxis should encourage healthcare institutions to adopt the IMPEDE score and NCCN-directed prophylaxis at least during the first 6 months after a diagnosis of multiple myeloma.
Future directives include performing a study focused on calculating an IMPEDE score and recommending NCCN-directed VTE prophylaxis at every treatment change in patients with multiple myeloma. This study will help identify which treatment for multiple myeloma results in more breakthrough VTE events and should analyze the efficacy of recalculating the IMPEDE score and the NCCN-directed VTE prophylaxis at each treatment change for multiple myeloma. Finally, a future study is needed to determine an efficient workflow within a clinical setting to accomplish pharmacy-led VTE risk stratification and VTE prophylaxis in patients with multiple myeloma.
Author Disclosure Statement
The authors have no conflicts of interest to report.
References
- Lyman GH, Carrier M, Ay C, et al. American Society of Hematology 2021 guidelines for the management of venous thromboembolism: prevention and treatment in patients with cancer. Blood Adv. 2021;5:927-974. Erratum in: Blood Adv. 2021;5:1953.
- Kristinsson SY, Pfeiffer RM, Björkholm M, et al. Arterial and venous thrombosis in monoclonal gammopathy of undetermined significance and multiple myeloma: a population-based study. Blood. 2010;115:4991-4998. Erratum in: Blood. 2011;117:3938.
- Bradbury CA, Craig Z, Cook G, et al. Thrombosis in patients with myeloma treated in the Myeloma IX and Myeloma XI phase 3 randomized controlled trials. Blood. 2020;136:1091-1104. Erratum in: Blood. 2020;136:1994.
- Palumbo A, Rajkumar SV, Dimopoulos MA, et al. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia. 2008;22:414-423.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): multiple myeloma. Version 1.2025. September 17, 2024. Accessed November 11, 2024. www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
- Dima D, Li A, Granat LM, et al. External validation of the SAVED score for venous thromboembolism risk stratification in patients with multiple myeloma receiving immunomodulatory drugs. Br J Haematol. 2023;201:280-284.
- Sanfilippo KM, Luo S, Wang TF, et al. Predicting venous thromboembolism in multiple myeloma: development and validation of the IMPEDE VTE score. Am J Hematol. 2019;94:1176-1184.
- Covut F, Ahmed R, Chawla S, et al. Validation of the IMPEDE VTE score for prediction of venous thromboembolism in multiple myeloma: a retrospective cohort study. Br J Haematol. 2021;193:1213-1219.
- Bravo-Perez C, Fernández-Caballero M, Soler-Espejo E, et al. Heparin versus aspirin thromboprophylaxis adds independent value to IMPEDE-VTE score for venous thrombosis prediction in multiple myeloma. J Thromb Thrombolysis. 2021;52:848-853.
- Bao L, Fang LJ, Xiao MY, et al. Validation of the IMPEDE VTE score for prediction of venous thromboembolism in Chinese patients with multiple myeloma: a single-center retrospective cohort study. Thromb Res. 2024;236:130-135.
- Piedra K, Peterson T, Tan C, et al. Comparison of venous thromboembolism incidence in newly diagnosed multiple myeloma patients receiving bortezomib, lenalidomide, dexamethasone (RVD) or carfilzomib, lenalidomide, dexamethasone (KRD) with aspirin or rivaroxaban thromboprophylaxis. Br J Haematol. 2022;196:105-109.
- Chakraborty R, Rybicki L, Wei W, et al. Abnormal metaphase cytogenetics predicts venous thromboembolism in myeloma: derivation and validation of the PRISM score. Blood. 2022;140:2443-2450.