Connecting Science to Practice
Asparaginase drugs have been a key part of improving outcomes
in the treatment of patients with acute lymphoblastic
leukemia (ALL). Use of these products is limited, however,
by their numerous adverse events, including thrombosis and
bleeding. Due to these risks, using anticoagulation with concurrent
asparaginase therapy is controversial. Patients with
ALL are at higher risk for thrombotic events, and anticoagulation
is an important supportive care measure for these patients.
This single-center retrospective study sought to evaluate
the incidences of thrombosis and bleeding among
patients receiving asparaginase to identify risk factors and
inform mitigation strategies. Our study provides information
on current research gaps and where future studies
should be directed.
Various risk factors have been identified for the presence of venous thromboembolism (VTE), such as type/stage of cancer, advanced age, obesity (body mass index [BMI] >30 kg/m2), presence of a central venous catheter, and history of a previous thrombosis.1-3 Patients with acute lymphoblastic leukemia (ALL) have been shown to be at higher risk for thrombosis, and the estimated 1-year cumulative incidence was 2.6%.2 Also, the incidence of VTE in adult patients with ALL (aged >30 years ) who were treated with asparaginase was estimated to be 42%.4 Specifically, Rank and colleagues found that the incidences were 3.7% in patients aged <10 years, 15.5% in patients aged 10 to 18 years, and 18% in patients aged 18 to 45 years.5 It is speculated that other factors, such as chemotherapy administration, may also contribute.1,2 In contrast to the evidence available regarding the occurrence of VTE in patients with ALL, data are limited regarding arterial thromboembolism (ATE). A review paper by Grover and colleagues summarized general risks associated with various chemotherapy and biologic agents and the occurrence of ATE, but they acknowledged that the literature is limited regarding many chemotherapy agents and risk for ATE.2 Furthermore, a study by Freyer and colleagues evaluated VTE occurrence with antithrombin administration as their end point and never evaluated the incidence of ATE specifically.3
It is thought that specific chemotherapy regimens put patients with cancer at higher risk for developing thrombosis.1,2 Asparaginase drugs include pegaspargase, calaspargase pegol, Erwinia asparaginase, and Escherichia coli asparaginase, and their use in patients with ALL has been shown to increase the risk for developing a thrombus.6-13 It is proposed that depletion of asparagine contributes to the increased risk.3,13,14 Asparagine is an amino acid component of antithrombin III (AT III). One study found that depletion of asparagine can lead to up to a 34% reduction in AT III concentrations when compared with baseline values.15 AT III is an anticoagulant and without adequate AT III concentrations, thrombin will not be inactivated and subsequently thromboses can develop.2,15 Higher doses of pegaspargase are associated with an increased incidence of adverse events (AEs), which includes thrombus, ultimately leading to changes in the recommended adult dosing from weight-based without a maximum dose to a maximum dose of 1 vial (3750 IU).14,16 Capping adult doses to 1 vial has been evaluated in clinical trials,3,14 and this is now recommended in the NCCN guidelines and was adopted at our institution.16 Conversely, bleeding is another known AE associated with asparaginase drugs because they cause depletion of clotting factors needed for thrombus formation.6-12 Despite the known risk for thrombus formation and bleeding with asparaginase drugs, it is unclear if patients should receive concomitant anticoagulation.3
This study evaluated the incidence of ATE and VTE and bleeding in patients with ALL who were receiving treatment with asparaginase drugs. Minimal data exist to identify additional risk factors that may contribute to either thrombotic or bleeding complications.17-19 Therefore, risk factors for the incidence of thrombosis and bleeding are investigated to help inform mitigation strategies.
Methods
This was a retrospective, single-center, observational study of patients initially treated for ALL with an asparaginase drug (pegaspargase or Erwinia asparaginase) between January 1, 2013, and July 31, 2021. Patients were identified via an electronic medical record (EMR) of asparaginase treatment and screened for inclusion. Patients were included if they received ≥1 dose of an asparaginase drug during the study time frame and were followed for ≥30 days after asparaginase administration. Patients were excluded if they received an asparaginase drug for refractory or relapsed ALL or if the information in the EMR was incomplete. This study was deemed exempt by the institution’s research subjects review board.
The primary objective of this study was to identify the cumulative incidence of thrombotic events in patients receiving treatment for ALL with pegaspargase or Erwinia asparaginase. Thrombotic events were defined as ATE, deep vein thrombosis (DVT), or pulmonary embolism (PE).
Secondary outcomes included the cumulative incidence of bleeding events and an assessment of thrombotic or bleeding events based on patient-specific factors, such as type of thrombosis, age, BMI, ALL type, or type of asparaginase drug received. Major bleeding events were identified from the EMR based on the International Society on Thrombosis and Haemostasis bleeding definition.20
The EMR was used for data collection. Patient demographics collected included age, sex assigned at birth, ethnicity, race, BMI, ALL type (B cell or T cell), National Cancer Institute risk group (standard risk, high risk, or unknown),21 asparaginase drug received, dose of asparaginase drug, if the dose was capped, and dates of administration to assess time to event.
Repletion with specific blood products, such as cryoprecipitate and AT III, was evaluated for all patients, given the routine practice of administering blood products and the potential contribution to the risk for thrombosis. Any administration within 14 days before a thrombotic event was deemed a potential contributing factor. For patients who had a thrombotic or bleeding event, additional laboratory values were collected at baseline, defined as within 72 hours from asparaginase administration, and at the time of the event. These included fibrinogen level, AT III level, platelet count, D-dimer level, prothrombin time, and activated partial thromboplastin time. Other data collected included the ALL treatment phase when the thrombosis or bleed occurred (induction or consolidation), type of thrombosis (ATE, DVT, PE), presence of central venous catheter, occurrence of major bleed, location of bleed, administration of anticoagulant, type of anticoagulant (eg, low-molecular-weight heparin [LMWH], unfractionated heparin, warfarin, direct oral anticoagulant), anticoagulant dosing (eg, treatment dose, prophylactic dose), and concomitant administration of antiplatelet agents or estrogen oral contraceptive.
The incidence of thrombosis or bleeding and time to event from the first asparaginase dose were collected throughout treatment and up to 30 days after the last dose of asparaginase. If multiple doses of an asparaginase drug were given within a 30-day time frame, the events were evaluated within 30 days of the last asparaginase dose. Thus, data were collected through August 31, 2021. The EMR was screened for key terms associated with thrombotic or bleeding events (thrombosis, thrombosis event, VTE, DVT, PE, bleed) to capture relevant clinic note documentation and imaging results reported. If multiple thrombotic events occurred, only the first occurrence was counted toward the incidence rate. If a patient had a relapse of ALL during first-line treatment, data collection stopped for that patient at the date of relapse.
Investigators collected data using a data dictionary and a standardized electronic data collection tool to reduce variability. All data abstractors were trained in data collection procedures by the senior investigator using example patients to demonstrate the retrospective EMR process and database input. In addition, 2 investigators performed a random sampling of 10% of collected data to evaluate accuracy. Date discrepancies were identified during the random sampling; therefore, all date variables (eg, asparaginase drug administration, thrombosis or bleeding event) for all patients received a secondary review, and discrepancies were reconciled. The senior investigator oversaw all data collection processes and assisted with any questions or clarifications regarding the data collection process or variables. The physician investigator reviewed and confirmed all thrombotic or bleeding events. Discrepancies were reconciled by exclusion if the physician was unable to confirm an event.
Statistical Analysis
Patient characteristics were summarized using medians and interquartile ranges for continuous variables, and counts and proportions for categorical variables. To account for varying lengths of follow-up, the incidence rate of thrombosis while on an asparaginase drug was calculated as the total number of thrombotic events (ATE, VTE, or PE) over the number of person-years of time on therapy. Person-years were defined as the time from a patient’s initiation on asparaginase until ≤30 days after the last asparaginase dose received. A patient was censored at the time of thrombosis, time of death, or time of relapse of ALL. At time of thrombosis, no subsequent asparaginase drug administrations counted toward at risk because we only considered first thrombosis as the event of interest. Incidence rates are presented with associated 2-sided 95% confidence intervals (CIs).
Secondary bleeding outcomes were analyzed similarly, and incidence rates are presented. Poisson models were used to identify patient and treatment factors that are associated with thrombosis or bleeding in this patient population. The Poisson model was adjusted for the varying lengths of follow-up. Incidence rate ratios were estimated and presented with associated 2-sided 95% CIs.
Results
A total of 138 patients received asparaginase drugs for the treatment of ALL from January 1, 2013, to July 31, 2021; 12 patients were excluded because they received asparaginase for the treatment of relapsed or refractory disease or had missing EMR information (Figure 1).
The remaining 126 patients were administered a total of 467 doses of asparaginase drugs, which included 416 doses of pegaspargase and 51 doses of Erwinia asparaginase. A total of 125 patients received pegaspargase with or without Erwinia asparaginase, and 1 patient received only Erwinia asparaginase during the study period. No doses of other asparaginase drugs were administered.
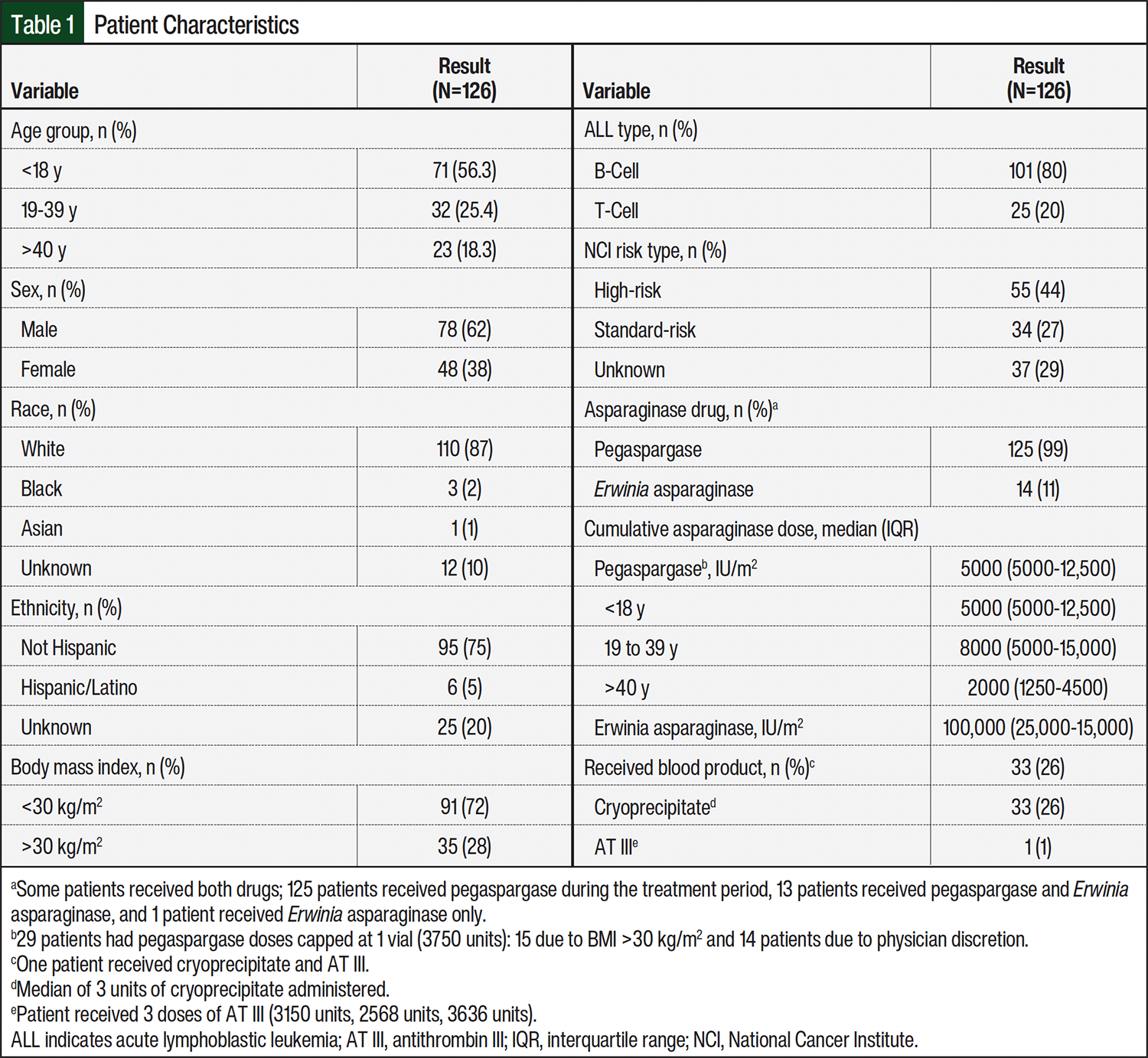
The study population had more patients aged ≤18 years (n=71) compared with patients aged >18 years (n=55). Most patients were White (87%) and not Hispanic or Latino (75%). A total of 28% were obese (BMI >30 kg/m2). The most common ALL type was B-cell leukemia (80%). Of the 126 patients, 33 (26%) received ≥1 unit of cryoprecipitate during treatment with an asparaginase drug; 1 patient received AT III repletion. Complete baseline characteristics are shown in Table 1.
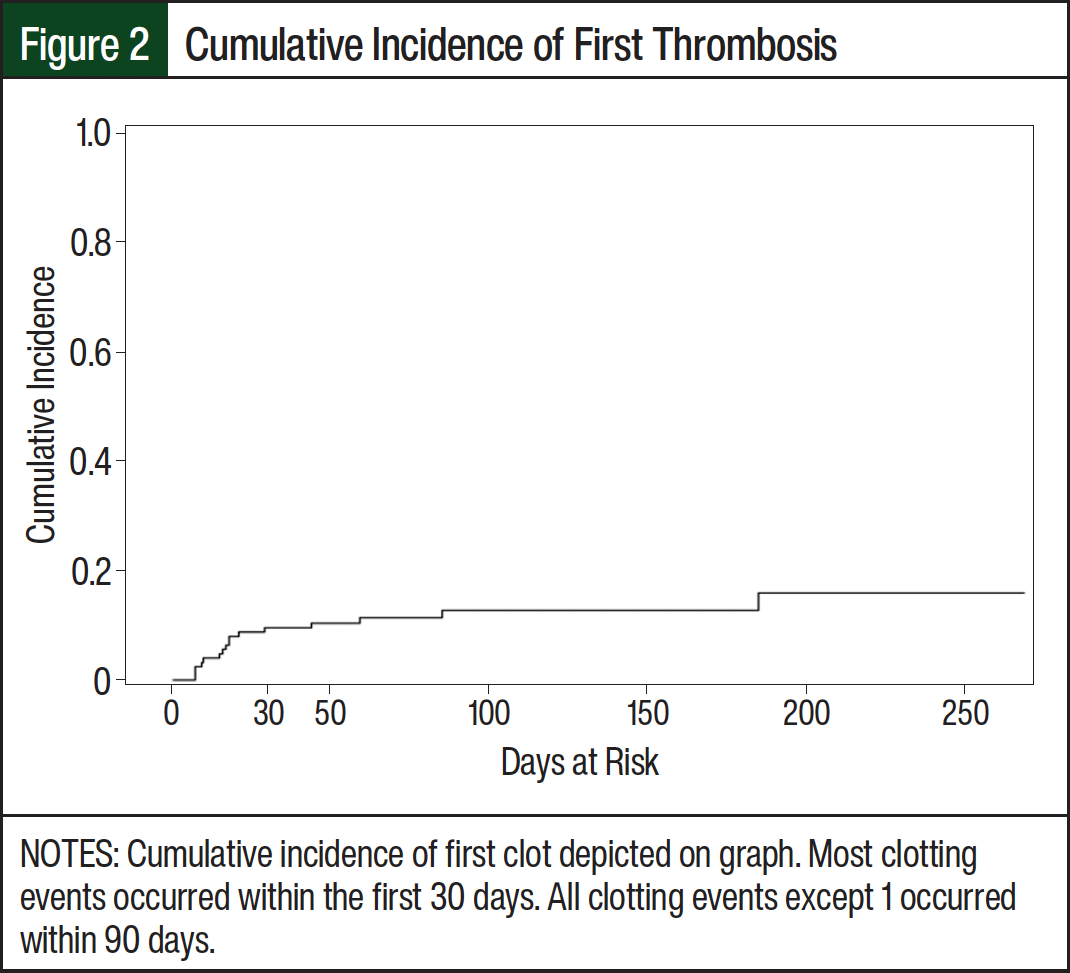
Sixteen (12.7%) of the patients receiving an asparaginase drug had a thrombotic event within 30 days of administration: 1 ATE, 13 DVTs, and 2 PEs. No thrombotic events occurred after administration of Erwinia asparaginase. The cumulative incidence of first thrombosis was 6 thrombotic events per 100 person-years at risk (95% CI, 0.84-42.7; Figure 2).
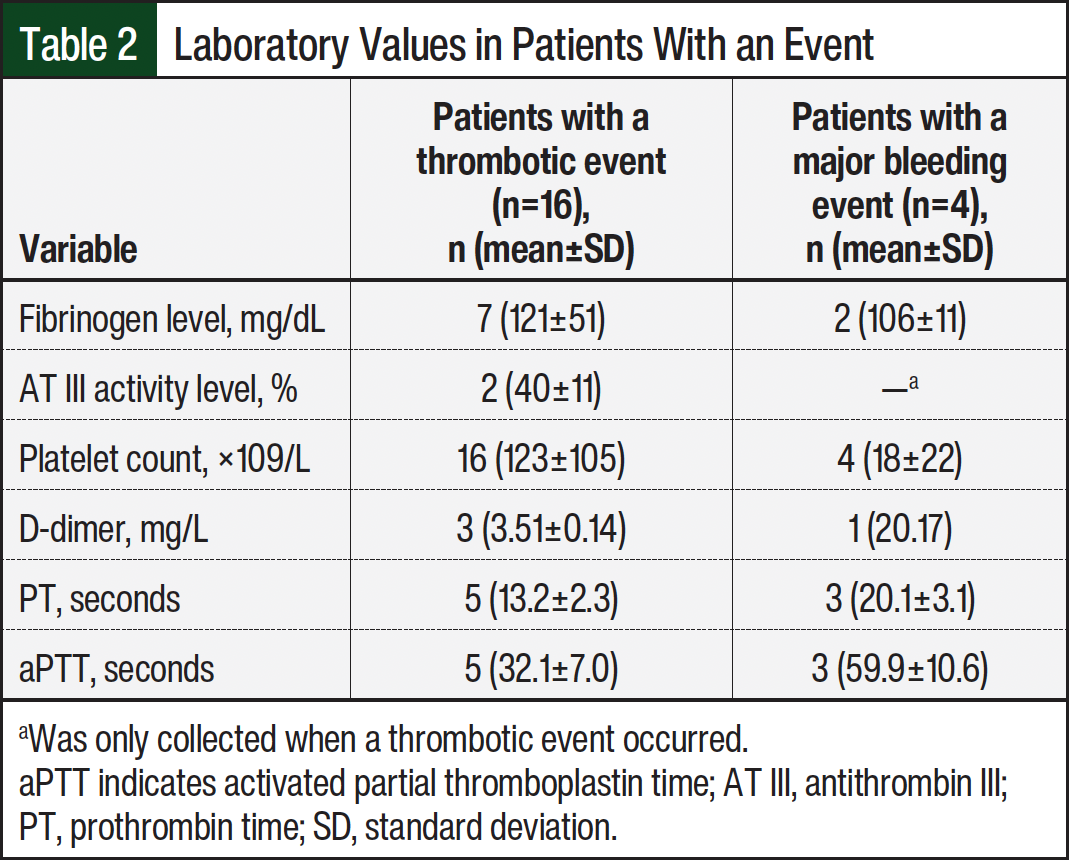
The cumulative incidence of thrombosis by day 30 was 9.5% (95% CI, 5.2-15.4). The median time from the first dose of asparaginase to thrombotic event was 17.5 days (range, 7-185), with most thrombotic events occurring during induction (12/16, 75%). The 1 ATE occurred at day 7, the DVTs occurred at a median of 17 days (interquartile range, 10-44), and both PEs occurred 18 days after administration. A total of 4 (3.2%) major bleeding events occurred. No bleeding events occurred after administration of Erwinia asparaginase. The cumulative incidence of first major bleed was unable to be evaluated because the number of events was too small for the Poisson model to converge when estimating the overall cumulative incidence rate of bleeding events. However, the cumulative incidence of major bleed by day 30 was 1.6% (95% CI, 0.3-5.1), with most (75%) bleeding events occurring during consolidation. Additional laboratory values collected for patients with a thrombotic or bleeding event are shown in Table 2.
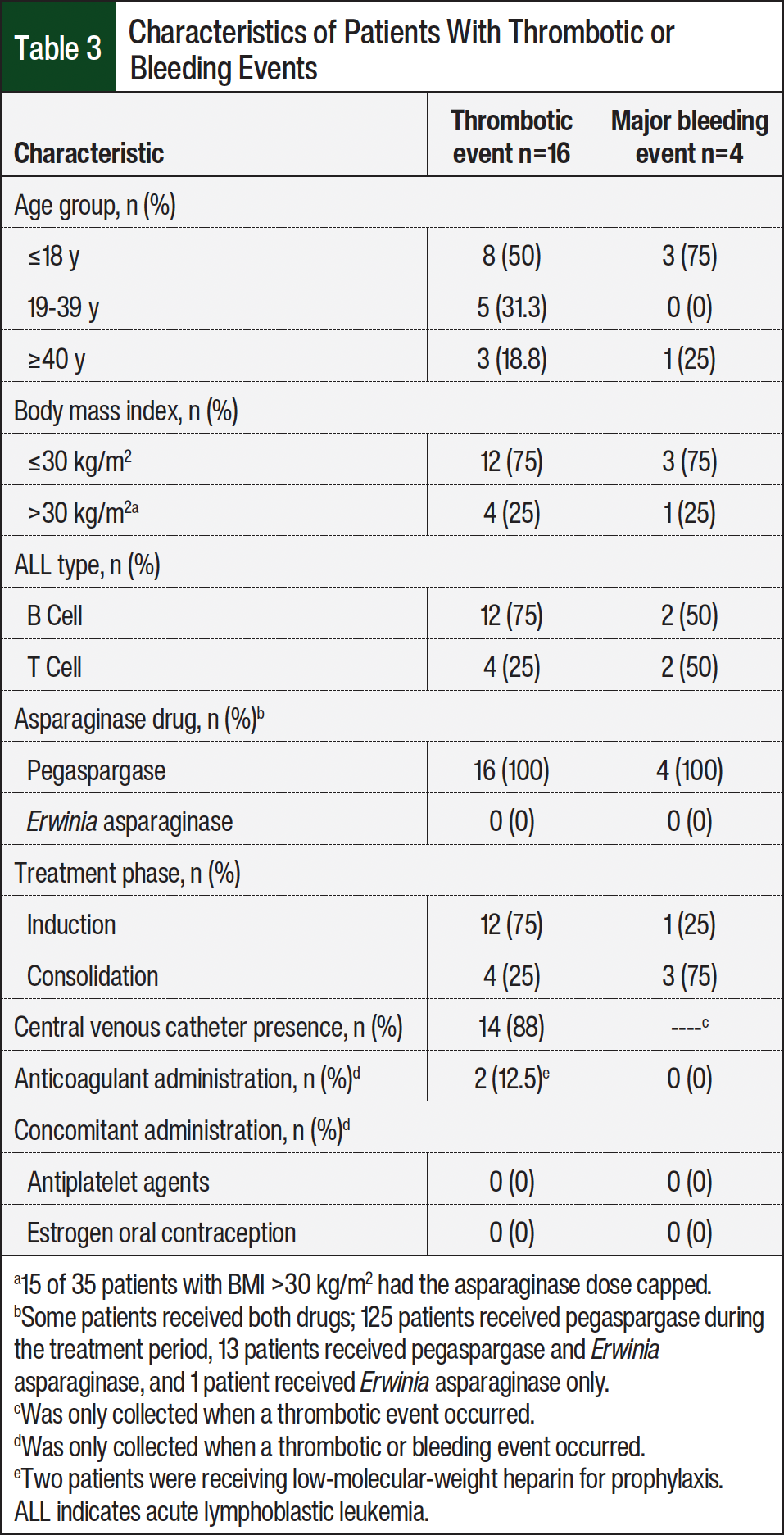
Of the 35 patients with BMI >30 kg/m2, 4 (11%) developed a thrombosis. Ten of 13 (77%) DVTs were associated with peripherally inserted central catheters. Four of 16 (25%) thrombotic events occurred within 14 days of cryoprecipitate administration and during induction, and 1 of these 4 patients had an ATE 1 day after receiving cryoprecipitate, and 2 of the 16 (12.5%) patients who had an event were receiving anticoagulation. When separated by event type (ATE, DVT, or PE) or patient characteristics (age, BMI, phase of ALL treatment, or asparaginase drug), the thrombotic event outcome was too small for the Poisson model to converge, and therefore, this secondary end point could not be evaluated. Only univariate analyses could be performed. No significant differences were found between those with an event and those without an event in age (thrombosis, P=.82; bleeding, P=.63), BMI (thrombosis and bleeding, P=1.00), or ALL type (thrombosis, P=.52; bleeding, P=.18). Thrombotic and bleeding events based on patient-specific factors are shown in Table 3.
Discussion
A thrombotic event occurred in 12.7% and a bleeding event in 3.2% of patients with ALL receiving ≥1 dose of an asparaginase drug for induction therapy over an 8.5-year evaluation. The cumulative incidence of first thrombosis was 6 thrombotic events per 100 person-years at risk (95% CI, 0.84-42.7). The incidence of a thrombotic event in the first 30 days of pegaspargase administration was higher than the incidence of a major bleeding event (9.5% vs 1.6%, respectively).
In the literature, the overall rate of thrombosis requiring anticoagulation reported with pegaspargase ranged from 5% to 27% compared with our rate of 12.7%.22 The majority of our thrombotic events were venous rather than arterial (94% vs 6%, respectively), and the majority (77%) of VTEs were DVTs associated with peripherally inserted central catheters. In 2 patients, thrombosis occurred while on prophylaxis anticoagulation. Given the lack of available data and high likelihood of thrombocytopenia in this patient population during induction, routine prophylaxis anticoagulation is not administered to these patients. AT III monitoring and repletion is also not routinely done because there is no published literature to our knowledge guiding this practice. If a patient has a thrombotic event and has received asparaginase, however, monitoring and repletion of AT III with heparin may be warranted. Further studies and guidelines are needed to better evaluate AT III repletion in patients receiving asparaginase and the administration of prophylaxis anticoagulation during induction, considering the risk for thrombocytopenia.
Patient-specific risk factors to evaluate for their association with thrombosis or bleeding risk (eg, age, BMI, ALL type, asparaginase drug used) were chosen based on results from previous literature.1-5 Although we attempted to combine pediatric and adult patients to increase the at-risk period to evaluate these associations, we were not able to use the Poisson model given the overall low event incidence and highly variable at-risk time across patients ranging from 7 to 269 days. Overall, pediatric patients (aged <18 years) accounted for 56% of the population in our study, and we found no numeric differences in thrombotic events based on age: 11% (aged <18 years), 15% (aged 19 to 39 years), and 13% (aged >40 years). Conversely, we found differences in bleeding events, with pediatric (aged <18 years) and adult patients (aged >40 years) having bleeding event rates of 4.2% and 4.3%, respectively, compared with no events in adolescent and young adults (AYAs; aged 19-39 years). These results, however, are difficult to interpret based on the overall low event rate and limited statistical analysis.
We found thrombotic events to be proportionally similar between obese (BMI >30 kg/m2) and non-obese patients, 11% and 13%, respectively. This is contrary to a previous study that found that obese patients were associated with higher risk for induction AEs, including thrombosis.16 These differences are likely due to our relatively small sample size of obese patients (n=35) and large pediatric population (n=71). This also may be due to the time frame of our study and the change in practice surrounding capping pegaspargase doses in patients with a BMI >30 kg/m2 and adjusting steroid dosing to pulse doses instead of continuous exposure. Overall, 29 patients had their pegaspargase doses capped at 1 vial (3750 units); 15 were due to BMI >30 kg/m2, which represents almost half of the obese patients (n=35) in our study. Most thrombotic events in our study occurred during induction (12/16, 75%), whereas the majority of bleeding events occurred during consolidation (3/4, 75%). This is similar to a previously published study indicating that the risk for the development of a thrombus is higher during the first cycle of chemotherapy.22 This is thought to be secondary to a higher hypercoagulable state during induction from more active leukemic disease, prolonged hospitalization, and higher steroid use during the first cycle.22
An interesting finding related to thrombotic events that occurred during induction and consolidation in our study was that cryoprecipitate administration seemed to have a greater impact on nonpediatric populations (adult and AYA). Four of 16 (25%) patients—1 adult and 3 AYAs—who had a thrombotic event also received cryoprecipitate repletion. Another study reported similar findings: 35% of patients who had thrombosis received cryoprecipitate replacement in the same cycle22; however, no studies, to our knowledge, compare age-groups. All 4 of our patients who received cryoprecipitate and had a thrombotic event had high-risk B-cell ALL, 3 of the 4 patients had a BMI >30 kg/m2, and all of the thrombotic events were during induction. Administration of cryoprecipitate is not standardized, but at our institution, it is typically reserved for patients with low fibrinogen levels, <50 to 75 mg/dL, or who are enrolled in clinical trials with a fibrinogen level <100 mg/dL. A total of 33 patients received ≥1 unit of cryoprecipitate during treatment with an asparaginase drug. Of these patients, 8 (24%) patients had a thrombotic event and 4 (12%) patients received cryoprecipitate within 14 days before the thrombotic event. Although the event rate is small, it illustrates the questionable benefit of the routine practice of repletion with cryoprecipitate during induction, particularly for adults and AYA patients.
Separately, concomitant use of anticoagulants or oral estrogen contraceptives can affect coagulopathy, and 2 patients who had thrombotic events were on LMWH; however, none of the bleeding events occurred while on anticoagulation. Overall, further studies are needed to determine the risk-benefit of cryoprecipitate administration and prophylaxis anticoagulation use in patients with ALL who are receiving an asparaginase drug.
Finally, when comparing asparaginase drugs, most patients received pegaspargase, and only 14 patients received Erwinia asparaginase. Based on our findings, it would be hard to draw conclusions regarding drugs and thrombotic risk given pegaspargase is typically the asparaginase drug given during induction, which is when the risk for thrombosis is the highest.
Strengths of this study include evaluating >100 patients spanning 8.5 years and reporting the incidence of thrombosis vis a vis a time-to-event evaluation and bleeding rates. ATE and VTE events were reported separately, which is not common in other reports. Nonetheless, our study has limitations. This was a retrospective study that relied on EMR data to collect variables and thrombotic and bleeding events, and it is possible that the actual number of events was underestimated or overestimated based on documentation. Still, these thrombotic and bleeding events are usually of high acuity and require medical intervention/documentation, making it unlikely that we significantly underestimated the occurrence. We included pediatric and adult patients and patients who received different asparaginase drugs to increase our sample size and the potential for a higher event rate to investigate associations between patient- or treatment-specific variables and events, but this may have caused heterogeneity in our population. As a result of the low event occurrence, the Poisson model could not be used to calculate cumulative incidence for secondary bleeding end points or to investigate associations based on patient- or treatment-specific factors, and only univariate analysis was performed. Furthermore, we did not collect data about the use of tyrosine kinase inhibitors in patients with Philadelphia chromosome–positive B-cell ALL, which may have provided additional information in those with or without a thrombotic event. We selected a 14-day cutoff for assessment of thrombosis after cryoprecipitate administration because we believed that this was a reasonable time frame in which the occurrence of a thrombosis may have been associated with cryoprecipitate administration. We recognize, however, that this time frame could be too long and not causative or too short and could have missed events. Last, we collected data for ≥30 days after the final asparaginase administration; and therefore, patients had different follow-up time frames, with some for only 30 days. Unfortunately, in those patients who did have events, we were unable to determine how these events affected the trajectory of their ALL treatment or doses administered, given the limited follow-up. This should be an area of focus for future research.
Conclusion
In patients who received asparaginase, the overall incidence of thrombotic events was higher compared with bleeding events. Most thrombotic events in patients receiving asparaginase drugs occurred during induction, whereas most bleeding events occurred during consolidation. No statistical differences were seen when evaluating age, BMI, ALL type, treatment phase, or type of asparaginase drug received between those who had a thrombotic or bleeding event and those who did not. One important finding is that almost one-third of thrombotic events occurring while receiving an asparaginase drug occurred in patients who had received cryoprecipitate repletion during induction treatment. Future studies are needed to examine the risk for cryoprecipitate repletion and examine the risk-benefit of AT III repletion and prophylaxis anticoagulation for patients receiving an asparaginase drug during induction.
Author Disclosure Statement
Moore is a consultant and on the Advisory Board of Beigene; Acquisto has received a grant from Melinta Therapeutics; Treptow is currently employed by Astellas Pharma but was employed by the University of Rochester Medical Center at time of data collection and review; Cendagorta, Lattuca, O’Dwyer, and Baran have no conflicts of interest to report.
References
- Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. Circulation. 2003;107(suppl 1):19-16.
- Grover SP, Hisada YM, Kasthuri RS, et al. Cancer therapy-associated thrombosis. Arterioscler Thromb Vasc Biol. 2021;41:1291-1305.
- Freyer CW, Carulli A, Ganetsky A, et al. Venous thromboembolism following pegaspargase in adults receiving antithrombin supplementation. Leuk Lymphoma. 2020;61:2200-2207.
- Grace RF, Dahlberg SE, Neuberg D, et al. The frequency and management of asparaginase-related thrombosis in paediatric and adult patients with acute lymphoblastic leukaemia treated on Dana-Farber Cancer Institute consortium protocols. Br J Haematol. 2011;152:452-459.
- Rank CU, Toft N, Tuckuviene R, et al. Thromboembolism in acute lymphoblastic leukemia: results of NOPHO ALL2008 protocol treatment in patients 1-45 years. Blood. 2018;131:2475-2484.
- Oncaspar (pegaspargase) injection, for intramuscular or intravenous use [prescribing information]. Servier Pharmaceuticals LLC; March 2024. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/103411s5207lbl.pdf
- Erwinaze (asparaginase Erwinia chrysanthemi) injection, for intramuscular or intravenous use [prescribing information]. Jazz Pharmaceuticals, Inc; December 2019. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2019/125359s098lbl.pdf
- Asparlas (calaspargase pegol-mknl) injection, for intravenous use [prescribing information]. Servier Pharmaceuticals LLC; November 2023. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2023/761102s013lbl.pdf
- Erwinase (asparaginase) injection, for intramuscular or intravenous use [prescribing information]. Jazz Pharmaceuticals, Inc; June 2020. Accessed September 22, 2025. www.drugs.com/pro/erwinase.html
- Kidrolase (L-asparaginase) injection, for intramuscular or intravenous use [prescribing information]. Jazz Pharmaceuticals; December 2017. Accessed September 22, 2025. https://pdf.hres.ca/dpd_pm/00042689.pdf
- Elspar (asparaginase) injection, for intravenous or intramuscular [prescribing information]. Lundbeck; July 2013. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2013/101063s5169lbl.pdf
- Rylaze (asparaginase Erwinia chyrsanthemi (recominant)-rywn) injection, for intramuscular use [prescribing information]. Jazz Pharmaceuticals, Inc; April 2024. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/761179s007lbl.pdf
- Truelove E, Fielding AK, Hunt BJ. The coagulopathy and thrombotic risk associated with L-asparaginase treatment in adults with acute lymphoblastic leukaemia. Leukemia. 2013;27:553-559.
- Wetzler M, Sanford BL, Kurtzberg J, et al. Effective asparagine depletion with pegylated asparaginase results in improved outcomes in adult acute lymphoblastic leukemia: Cancer and Leukemia Group B Study 9511. Blood. 2007;109:4164-4167.
- Homans AC, Rybak ME, Baglini RL, et. al. Effect of L-asparaginase administration on coagulation and platelet function in children with leukemia. J Clin Oncol. 1987;5:811-817.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): acute lymphoblastic leukemia. Version 2.2025. June 27, 2025. Accessed January 26, 2024. www.nccn.org/professionals/physician_gls/pdf/all.pdf
- Stock W, Luger SM, Advani AS, et al. A pediatric regimen for older adolescents and young adults with acute lymphoblastic leukemia: results of CALGB 10403. Blood. 2019;133:1548-1559.
- Advani AS, Larsen E, Laumann K, et al. Comparison of CALGB 10403 (Alliance) and COG AALL0232 toxicity results in young adults with acute lymphoblastic leukemia. Blood Adv. 2021;5:504-512.
- Xu L, Wei X, Wei Y, et al. Coagulation disorder after treatment of pegaspargase and L-asparaginase. Blood. 2015;126:4876.
- Schulman S, Kearon C. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. 2005;3:692-694.
- Schultz KR, Pullen DJ, Sather HN, et al. Risk- and response-based classification of childhood B-precursor acute lymphoblastic leukemia: a combined analysis of prognostic markers from the Pediatric Oncology Group (POG) and Children's Cancer Group (CCG). Blood. 2007;109:926-935.
- Aldoss I, Douer D. How I treat the toxicities of pegasparaginase in adults with acute lymphoblastic leukemia. Blood. 2020;135:987-995.